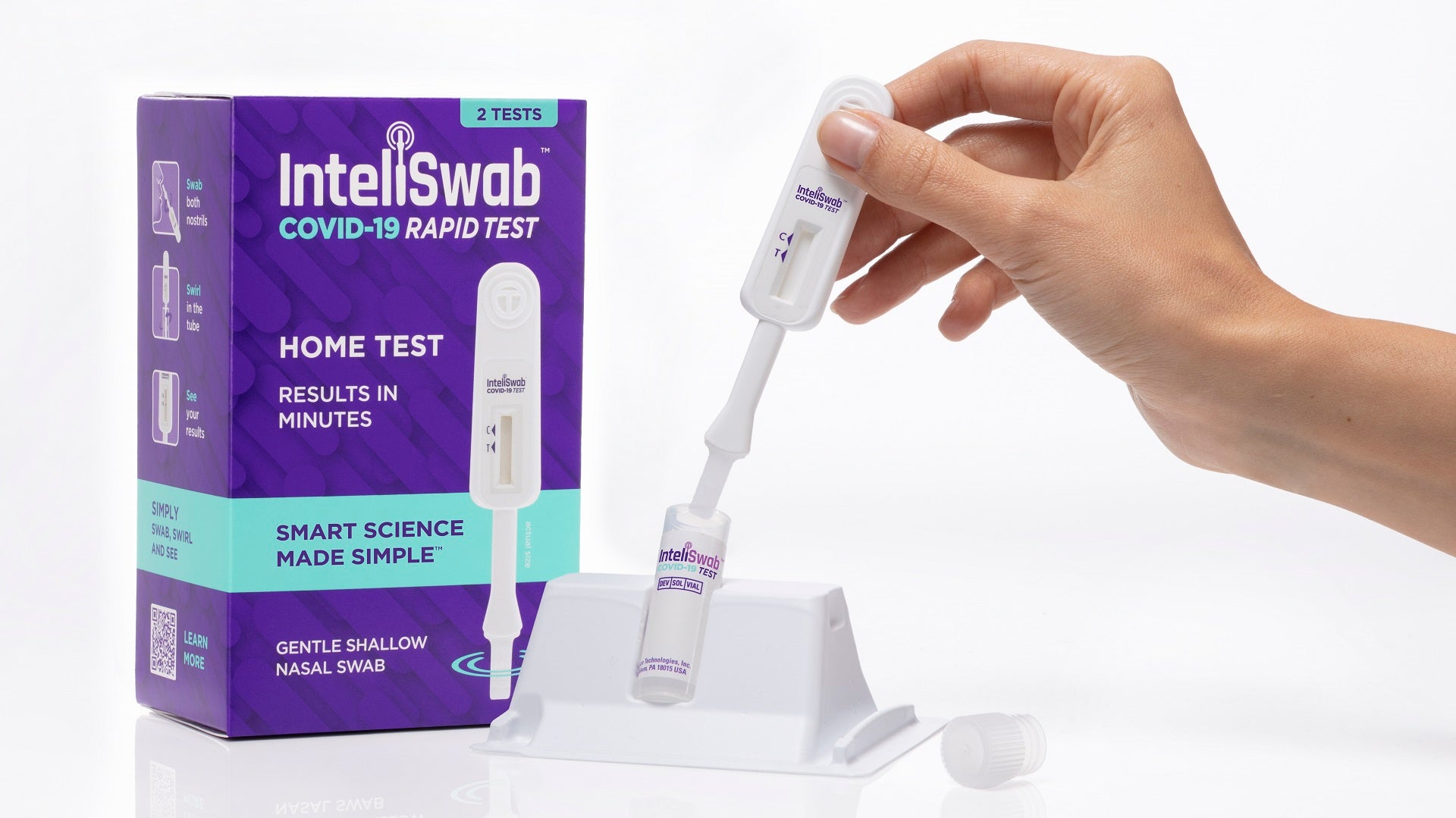
The US Food and Drug Administration (FDA) has authorised OraSure Technologies’ InteliSwab COVID-19 Rapid Tests for use in children aged two to 14 years.
Previously, the tests were authorised for use in adults and children aged 15 to 17 years when given by an adult.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The InteliSwab COVID-19 Rapid Test was authorised by the regulator for use without a prescription. The InteliSwab COVID-19 Rapid Test Rx also secured approval for home use with a prescription.
The FDA also granted authorisation for the InteliSwab COVID-19 Rapid Test Pro for professional use in point of care (POC) settings.
OraSure has conducted trials to assess the test’s performance, usability and tolerability in children as part of its submission to the regulator for expanded authorisation.
The InteliSwab test showed an 85% positive percent agreement and 98% negative percent agreement from a combined performance perspective in children aged two and above.
Including paediatric users, its overall accuracy is 93%, which is a similar accuracy rate compared to studies in adults.
OraSure Technologies president and CEO Stephen Tang said: “Helping our children stay in school and get back to normal is crucial to their wellbeing and continued development.
“Fast, frequent testing is a key component to making this happen and we are delighted that our InteliSwab test can now be used for children.”
Additionally, the company has launched InteliSwab Connect, a new app to report test results to public health authorities.
Available on the Apple App Store and through Google Play, the new app will help employers track the prevalence of Covid-19 in the workplace.





