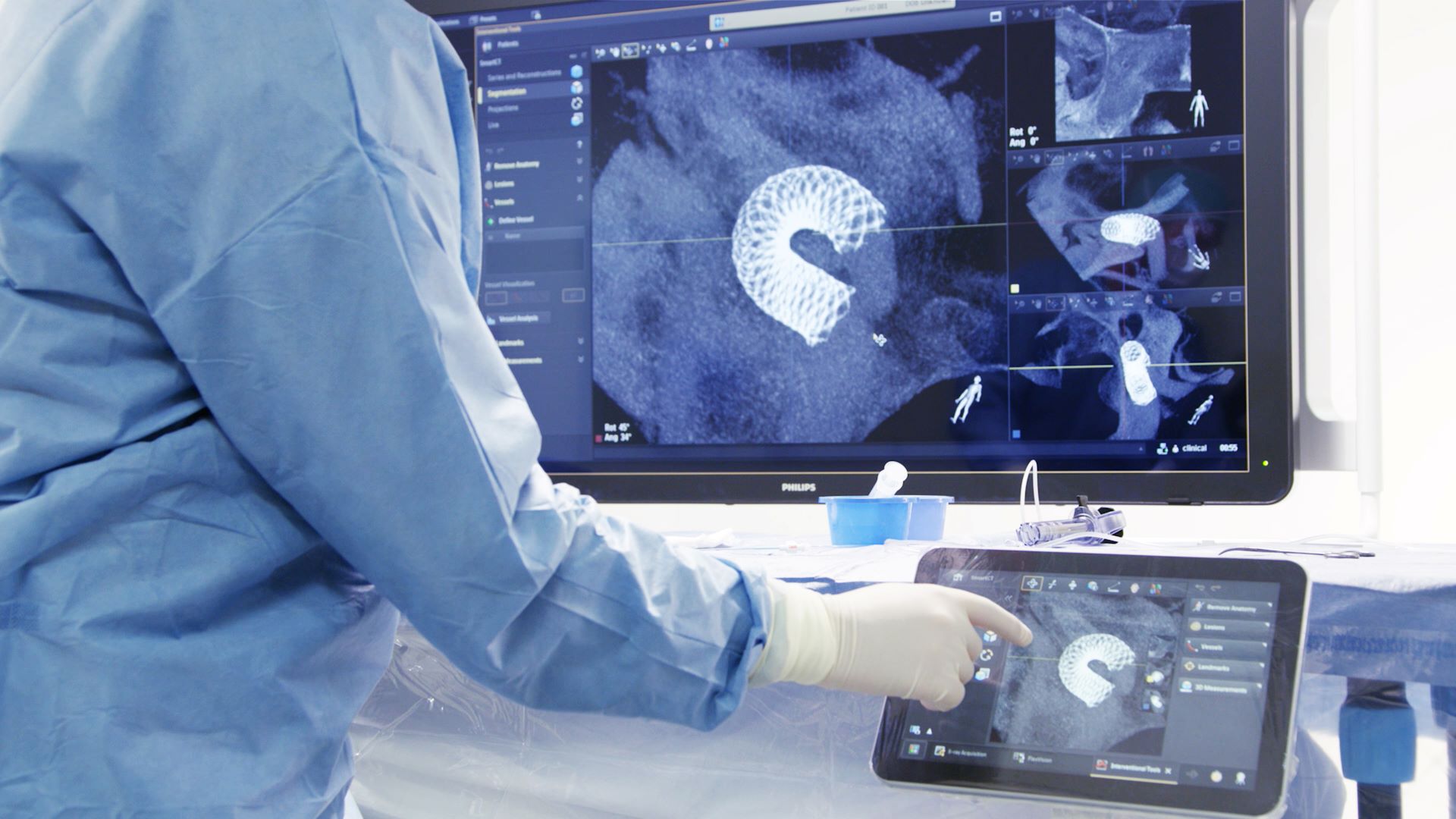
The US Food and Drug Administration (FDA) has granted 510(k) clearance to Philips’ SmartCT image acquisition, visualisation and measurement software.
A crucial part of the Philips Azurion Image Guided Therapy System, SmartCT will offer interventionalists CT-like 3D images (Cone Beam CT), which will aid in the diagnosis, therapy planning, treatment and follow-ups for interventional radiology procedures.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
SmartCT can improve clinical confidence, streamline workflows and boost productivity by bringing the intuitive touchscreen controls of advanced 3D image acquisition, visualisation, vessel/organ segmentation and quantitative measurements inside the sterile zone of the interventional lab and to the table-side.
Furthermore, with software applications for angiography, neurology, soft-tissue imaging and guidewire/catheter navigation, SmartCT can support various procedures, including treatment of aneurysms, vascular diseases and liver tumours.
Philips image guided therapy systems general manager Ronald Tabaksblat said: “A key part of our image-guided therapy strategy is to combine high-quality, low X-ray dose imaging with a superior user experience that allows interventional radiologists to diagnose and treat patients as part of smoother, safer and less interrupted workflows.
“Philips SmartCT is a major step forward in 3D imaging, enhancing confidence in the interventional suite and supporting key elements of the quadruple aim of better patient outcomes, enhanced patient and staff experiences and lower cost of care.”
The Azurion platform combines the tools required for complex interventional procedures with vital lab systems for an uncluttered lab environment.
SmartCT provides total control of the Azurion platform with a touchscreen tablet located next to the interventional radiology table, allowing clinicians to remain inside the sterile zone rather than using an adjacent control room.
Furthermore, it will help to reduce disruptions to procedures, save time and permit interventionalists to stay focused on patients while making quick and well-informed decisions.
In November last year, Philips expanded its home care portfolio for chronic obstructive pulmonary disease patients with the launch of a non-invasive Philips Ventilator BiPAP A40 EFL.





