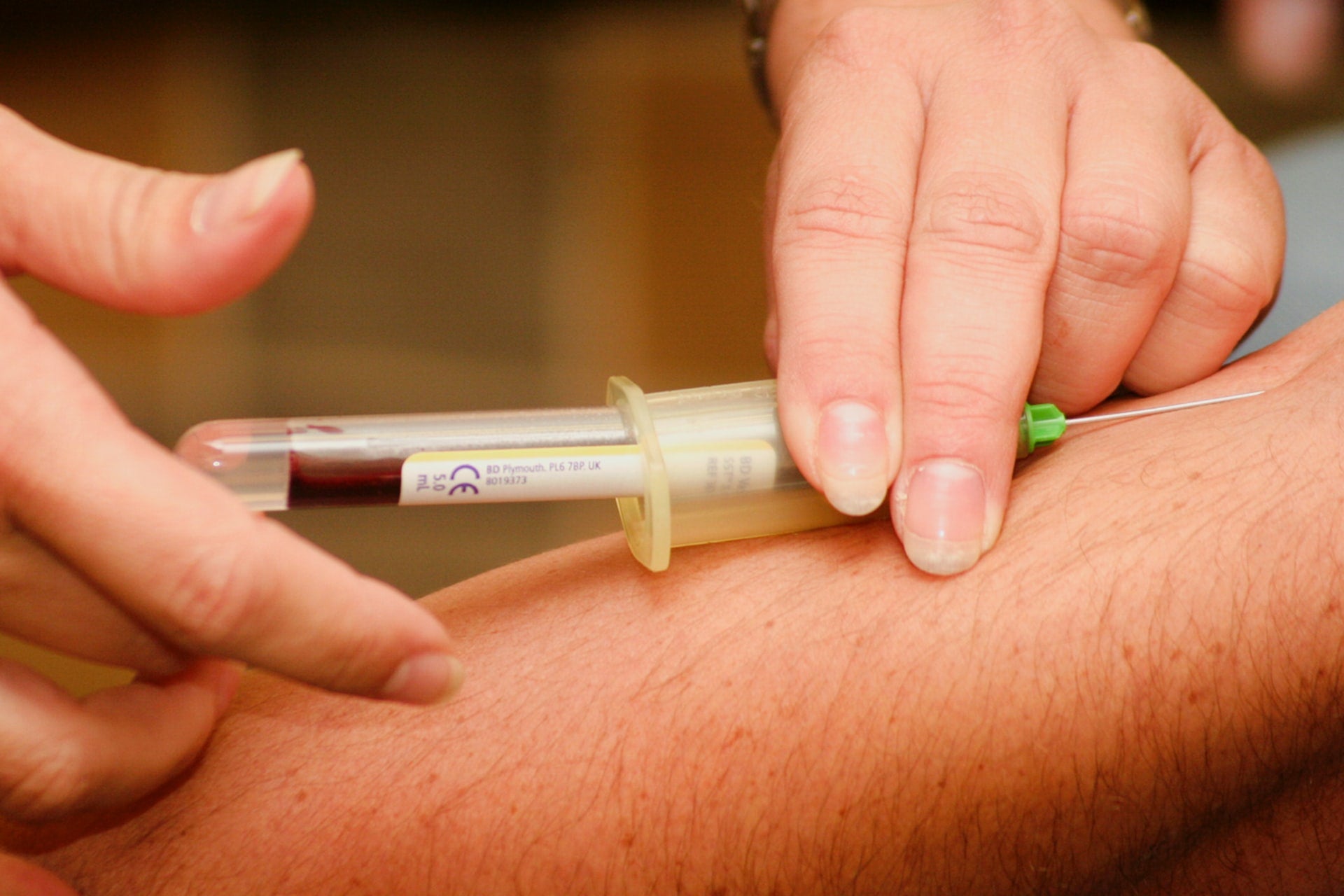
The US Food and Drug Administration (FDA) has approved the Qiagen Liaison QuantiFeron-TB Gold Plus assay for use on DiaSorin’s Liaison XS platform.
The decision comes approximately two years after the FDA approved the commercial launch of the Liaison QuantiFeron-TB Gold Plus assay. The assay has been available on LIAISON XL analyser platforms in the US since 2019.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
An interferon-gamma release assay (IGRA), LIAISON QuantiFERON-TB Gold Plus was developed by Qiagen and DiaSorin to streamline laboratory automation for latent tuberculosis (TB) screening.
QuantiFERON-TB looks for interferon-gamma from T-cells that have come across TB bacteria.
The latest regulatory approval from the FDA increases the accessibility of automation solutions for processing the Qiagen blood-based assay for latent TB detection in the US.
It will also support moving from traditional tuberculin skin tests to modern blood-based tests for the identification of latent TB in patients.
The addition of the fully automated LIAISON XS platform will also broaden the range of potential customers to include small healthcare clinics as well as larger hospitals, medical centres and reference laboratories.
Qiagen CEO Thierry Bernard said: “QuantiFERON-TB Gold Plus continues to set new standards in the global fight against TB, a disease that remains a persistent killer and impacts people around the world.
“Our partnership with DiaSorin has enabled customers to absorb the increasing demand for TB detection with access to a proven automation solution on the LIAISON XL platform, especially in larger reference labs and hospitals.
“The addition of the LIAISON XS platform will open up new joint opportunities for us to reach new customer segments requiring lower-throughput options.”





