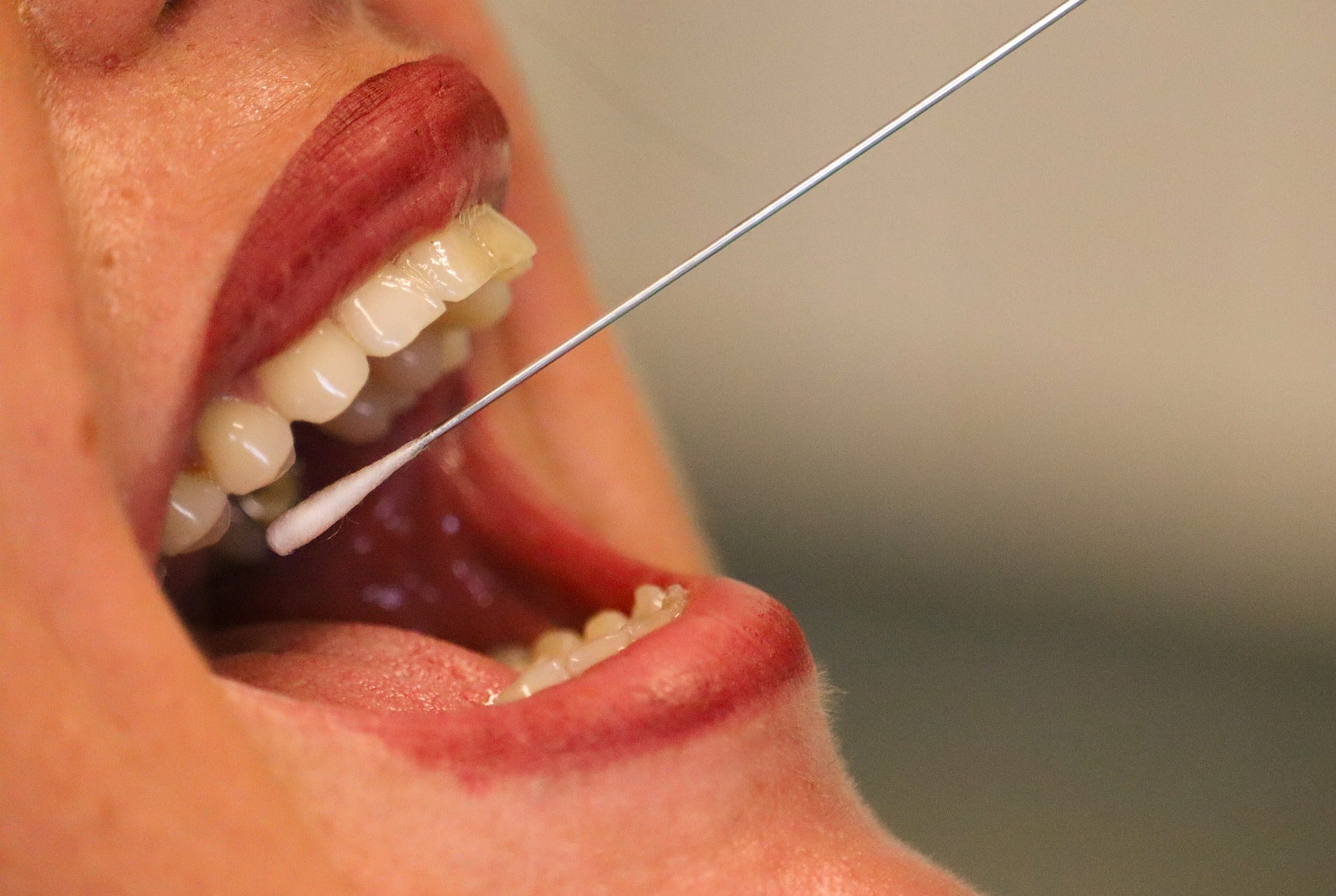
The US Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for Thermo Fisher Scientific’s new saliva sample collection method for its automated Covid-19 testing system.
Thermo Fisher received the authorisation to run Covid-19 tests on the Amplitude Solution using saliva samples collected with the Spectrum Solutions SpectrumDNA SDNA-1000 collection device.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The self-contained SDNA saliva collection device provides critical sample consistency while neutralising DNA/viral RNA transcripts after collection of samples to help ensure accurate test results.
It is a single device for DNA/viral RNA biosample collection, transportation and storage.
The device reduces costs associated with sample failures and recollection.
The Amplitude Solution is a new automated molecular testing solution, which has the capability to analyse up to 8,000 Covid-19 samples in 24 hours.
It helps laboratories to scale Covid-19 polymerase chain reaction (PCR) testing to reduce the spread of the disease and restore communities and economies.
With minimal time, equipment and staffing, the solution offers maximum throughput and empowers clinical labs to expand testing capacity.
Thermo Fisher Scientific genetic testing solutions senior medical director Manoj Gandhi said: “With the increase of Covid-19 cases due to more transmissible variants such as the Delta variant, routine testing for SARS-CoV-2 remains an effective strategy to curb the pandemic.
“By enabling labs and public health officials with additional flexibility in sample collection on a high-throughput system, we are improving their ability to meet the increased demand which helps to monitor the spread of Covid-19 and, eventually, save lives.”
The Amplitude Solution’s high-throughput capabilities help to address the testing requirements for communities, universities, companies and schools to return to pre-pandemic life.
Last month, the company launched the new Thermo Scientific SpeciMAX Stabilized Saliva Collection Kit, which provides a simple method for saliva collection.





