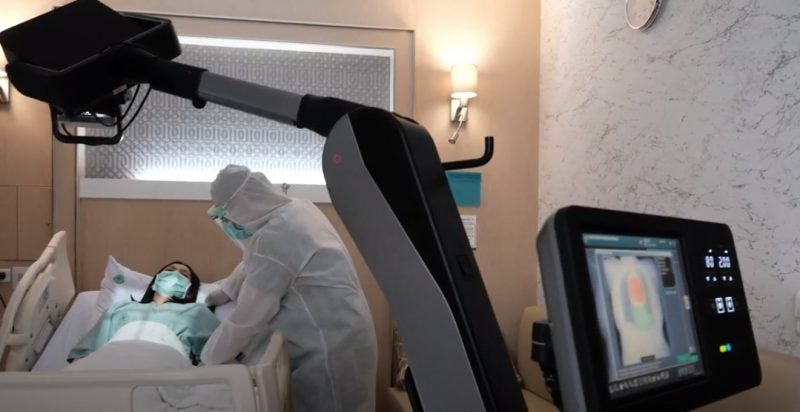
Fujifilm has introduced its artificial intelligence (AI)-powered chest X-ray software, CXR-AID, in Japan following approval from the Pharmaceuticals and Medical Devices Agency (PMDA).
Developed using Lunit’s AI technology, CXR-AID can identify major abnormal findings including chest nodule, consolidation and pneumothorax from X-ray images.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
These findings are visualised in colour as a heatmap display, including each score.
Leveraging the software, doctors and medical specialists can lower missed cases and efficiently diagnose chest diseases from the X-ray.
Offered along with Fujifilm’s SYNAPSE picture archiving and communication system (PACS), CXR-AID will be distributed across hospitals and medical centres in the country for improved diagnosis, the company noted.
Fujifilm medical systems business IT solutions division senior manager Naoya Yamakawa said: “We try to listen to our customers’ voice and Lunit’s AI software is gaining very positive feedback from the actual users across the world.
“For example, one of our customers, Salud Digna, known as the largest medical institution of Mexico, uses our AI-embedded PACS to analyse more than 800,000 X-ray images per year.”
In 2018, Fujifilm entered into a partnership with Korean medical AI company Lunit and Salud Digna.
Lunit offers deep-learning-based AI technology, called Lunit INSIGHT CXR, that assesses chest X-rays.
The AI products integrating Lunit AI to Fujifilm’s PACS system and X-ray devices are marketed globally.
The companies are also extending the scope of their partnership.
Lunit AI is used in Fujifilm’s FDR nano mobile X-ray device, FDR Xair portable X-ray devices, FDR Smart X General X-ray device. It is being utilised in clinical settings to identify Covid-19 symptoms and tuberculosis in areas with reduced availability of doctors.
The products are installed globally, including in the UK, Thailand, Vietnam, Turkey, the Middle East and Africa.
In March, Fujifilm Europe launched its rapid antigen test kit for the detection of SARS-CoV-2 infection, following the receipt of the CE certificate.





