GE HealthCare has received 510(k) clearance from the US Food and Drug Administration (FDA) for its new deep learning-based image processing software, Precision DL.
As part of the company’s Effortless Recon DL portfolio, the new software offers image quality performance benefits that are generally linked with hardware-based Time-of-Flight (ToF) reconstruction.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
These benefits include improved quantitative accuracy, contrast-to-noise ratio and contrast recovery.
GE HealthCare imaging president and CEO Jan Makela said: “Clinicians are seeing the value of applying deep learning technology to enhance image quality with our multi-modality family of Effortless Recon DL applications, which already includes AIR Recon DL for MR, TrueFidelity for CT and Helix DL for X-ray.
“Now we are proud to add Precision DL for PET/CT, enabling more precise and personalised care across healthcare systems’ imaging departments.”
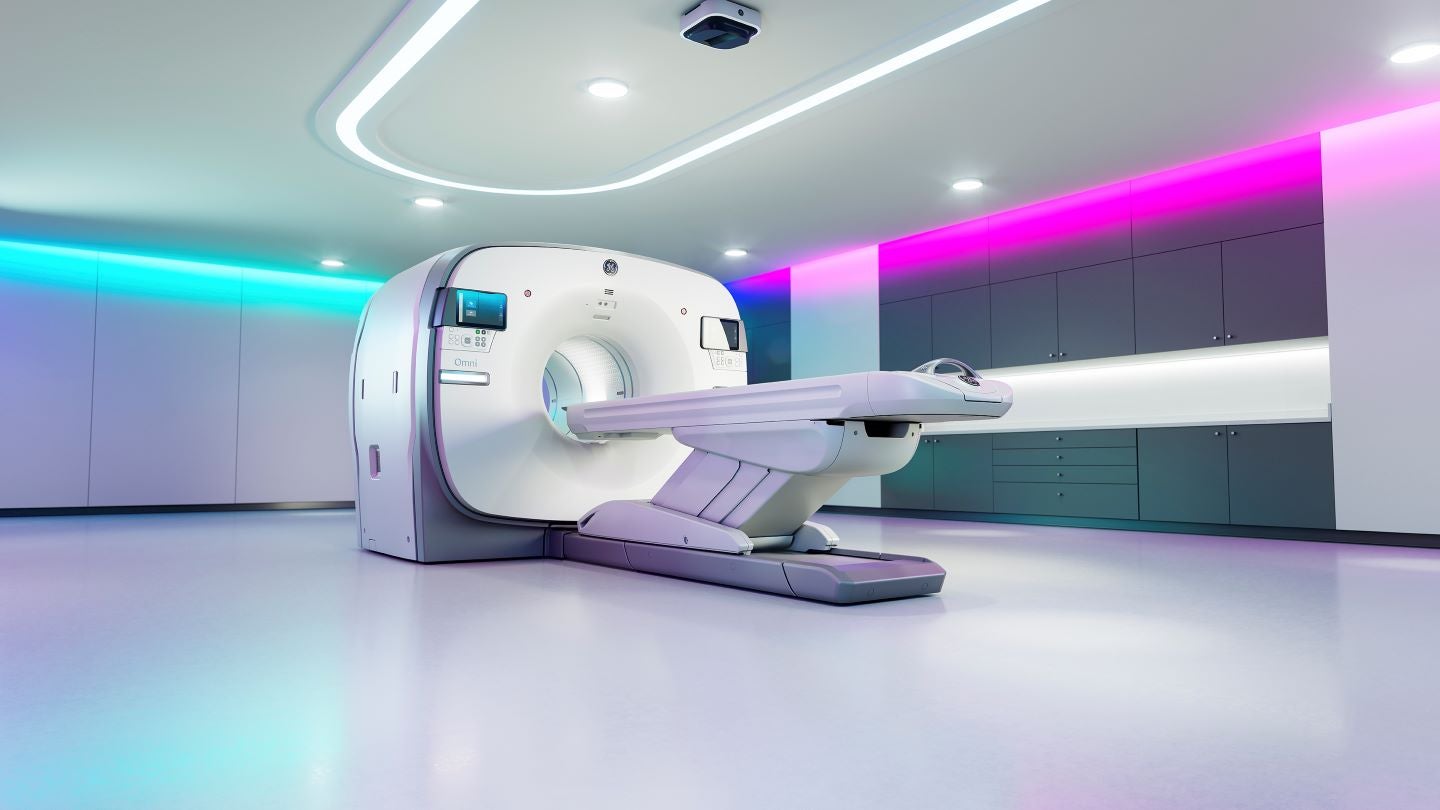
The company’s PET/CT, Omni Legend, offers artificial intelligence-based Precision DL technology.
The combination of Precision DL and Omni Legend’s sensitive third-generation digital detector technology allows clinicians to decode coincidence events at fine resolutions to enable informed diagnoses and treatment planning.
It also paves the way for shifting away from ToF technology and advancing to the next generation of PET/CT performance.
Precision DL leverages an advanced deep neural network, which underwent training using thousands of images produced through multiple reconstruction methods, including ToF reconstruction.
According to the company, the new software offers on average a 23% improvement in contrast-to-noise ratio; a 42% increase in small, low contrast lesion detectability and an 11% improvement in contrast recovery.
The Precision DL system also improves feature quantification accuracy by 14%.





