Polish biotech company GeneMe has developed FRANKD, a fast, scalable mouth swab test for Covid-19.
The test can be completed on-premises in an average of 13-25 minutes and does not require any laboratory involvement.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The GeneMe FRANKD Covid-19 test has shown to have 100% sensitivity and 100% specificity by the Gdańsk University of Technology.
FRANKD provides results in less time than the International Health Organization(WHO)-recommended gold-standard reverse transcription polymerase chain reaction (RT-PCR) reference test.
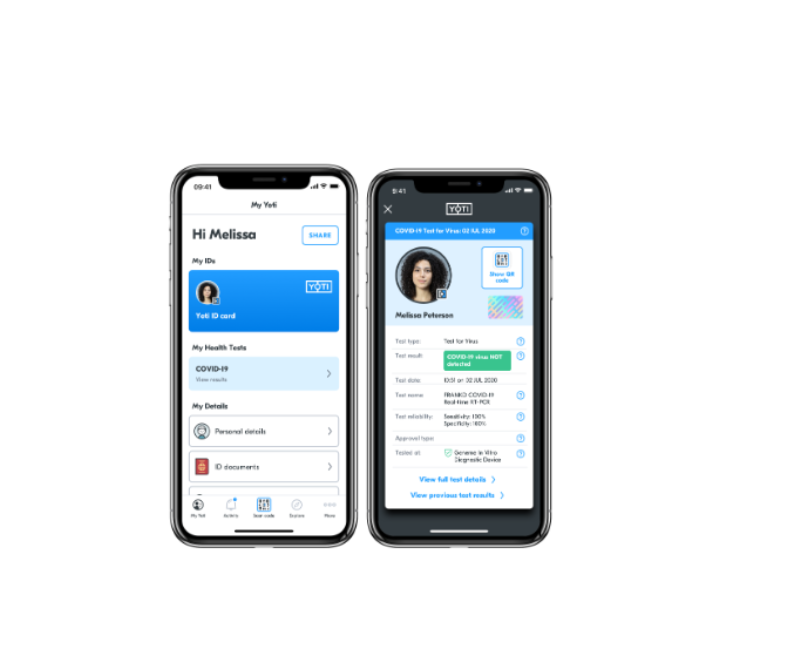
With approval, results are automatically issued to each individual’s secure, free digital Yoti ID app, a digital ID and credential management platform.
The company said that the test will cost £10 for businesses in the region, making it cost-effective to carry out daily on a mass scale and for point-of-entry testing at care homes, airports, offices and several other locations.
GeneMe founder and CEO and FRANKD developer Dawid Nidzworski said: “We are extremely pleased with the quality of the FRANKD test and the contribution it will make towards the fight against the coronavirus pandemic and its damaging effects on public health and society.”
Currently, the company is holding discussions with governments and businesses across Europe to introduce millions of tests in the next few months.
Unlike the scarce reagents used in RT-PCR tests, FRANKD test uses a patented enzyme to quickly produce a reliable result.
FRANKD is said to be scalable with five million monthly tests expected from August.
Due to the use of a portable machine analyser to amplify the virus within the sample, high test accuracy is expected to become possible.
The company said that 90 people can be tested at a time, every 30 minutes with one device, significantly reducing the testing cost.
The company expects European CE regulatory certification by early June.
Due to a specific web application, all 90 tests within a batch will be uniquely assigned to the correct owner prior to entering the analyser machine.
Currently, Yoti app is available in English. The app in French, Spanish, Polish and German will be released shortly.





