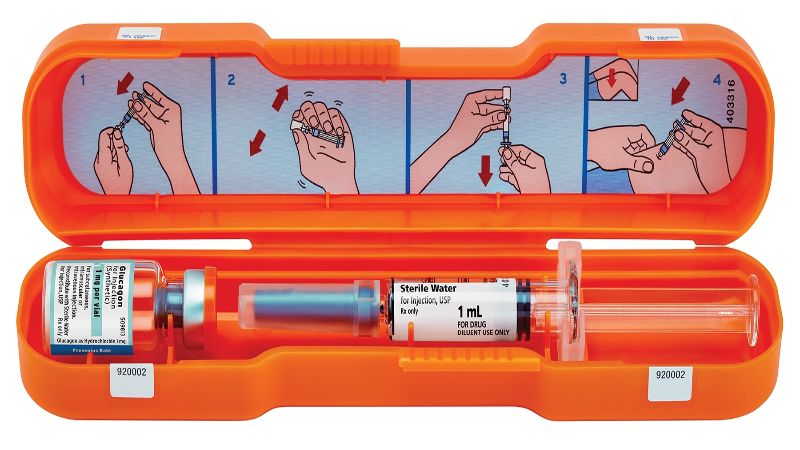
German healthcare company Fresenius Kabi has introduced its Glucagon Emergency Kit in the US to treat sudden hypoglycaemic episodes in diabetic patients.
The FDA-approved kit comprises Glucagon for Injection 1mg along, a prefilled glass syringe and 1ml of sterile water for injection.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Easily portable, the kit will enable immediate administration of the glucagon injection, if the patient suffers a severe hypoglycaemic attack. It also comes in a bright orange case to make it easier to find in case of an emergency.
Hypoglycaemia is a condition when the blood sugar (glucose) level of the body drops to a low level.
Glucagon for Injection is an anti-hypoglycaemic agent, which treats the condition in paediatric and adult patients with diabetes. However, it is not for use in patients with pheochromocytoma, insulinoma, or who are hypersensitive to glucagon.
Fresenius Kabi USA president and CEO John Ducker said: “For people with diabetes, a severe hypoglycemic episode can occur anywhere and at any time.
“Our new Glucagon Emergency Kit allows clinicians flexibility and choice for treating patients experiencing severe hypoglycemia. This is another example of Fresenius Kabi’s commitment to providing cost-effective products that deliver life-saving care.”
The company will also provide injection training kits to healthcare providers to help them to train patients.
A copay assistance programme is also in place to make the kit affordable to eligible patients.
As a global healthcare firm, Fresenius Kabi delivers medicines and technologies for infusion, transfusion and clinical nutrition.
The company’s US headquarters is in Lake Zurich, Illinois.





