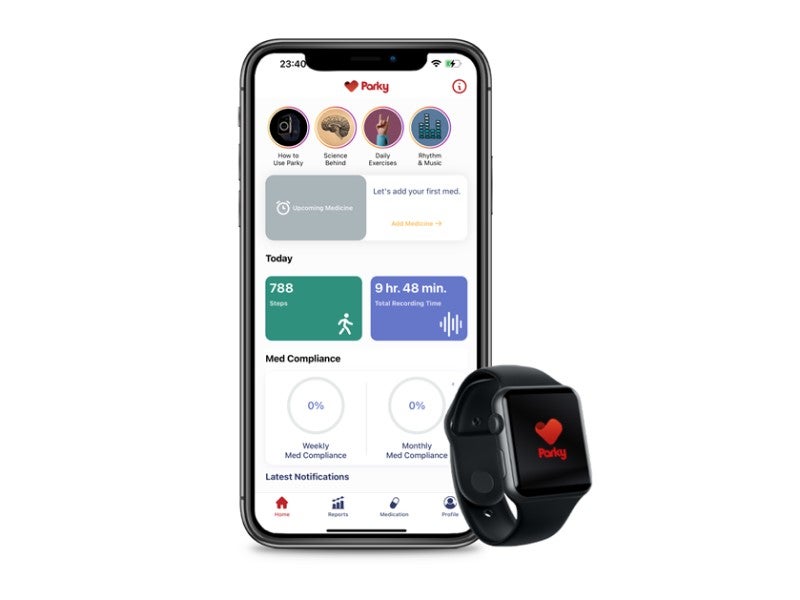
h2o therapeutics has secured 510(k) clearance from the US Food and Drug Administration (FDA) for Parky, a prescription mobile app designed to monitor Parkinson’s Disease.
The Apple Watch-based Parky app has been designed for monitoring tremor and dyskinesia symptoms of Parkinson’s Disease patients in real time.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is a tool intended for meaningful and reliable data sharing between patients and medical specialists, which will enable tracking of the disease’s course.
The mobile app helps monitor Parkinson’s Disease patients and manage their treatment procedures more accurately.
It also provides medicine reminders and produces symptom reports, to be shared with selected healthcare professionals, in real time.
The app uses a tool developed by Apple, known as Movement Disorder API, which was validated in a study conducted with 343 participants.
h2o therapeutics stated that the Parky app assists healthcare providers in developing a clinical profile of the patient outside of the clinic.
Reinforcing the possibility of data-driven, tailored treatment procedures, the app helps cover the gap between in-clinic settings and real life.
h2o therapeutics founder Yagmur Selin Gulmus said: “As a woman-founded, non-VC-backed company based in Turkey, receiving our first 510(k) clearance is a huge milestone for us.
“We believe Parky will bring great value to the Parkinson’s Disease community as an easily scalable and data-driven product.”
Additionally, the company has two other digital therapeutics products based on wearable devices in the pipeline. It plans to complete FDA submissions for the products next year.
According to the company, approximately ten million patients across the globe are living with Parkinson’s Disease, which is said to be the fastest-growing neurological disorder.





