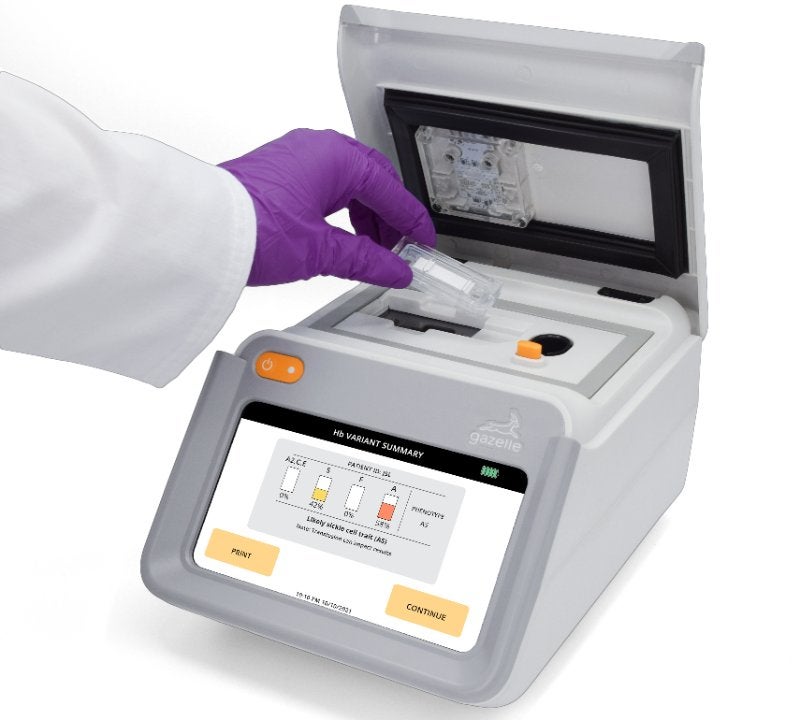
US-based health technology start-up Hemex Health has upgraded its Gazelle Hb Variant test.
The latest enhancement will enable more precise measurements of foetal haemoglobin Hb F and help in the monitoring of hydroxyurea therapy in point-of-care settings.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This development comes after the company released another enhancement to enable a Hb Variant test that detects beta thalassemia disease and trait as well as sickle cell disease (SCD) and trait.
Hemex Health CEO Patti White said: “Gazelle continues to deliver unprecedented capability to populations with a high prevalence of sickle cell and beta thalassemia.
“In addition to this Hb F measurement improvement, additional features are planned for Gazelle that will improve healthcare for sickle cell and beta thalassemia patients who have not had access to this level of testing.”
Hydroxyurea has been used to treat adult SCD since the 1980s.
The medication was also recently approved for children by the US Food and Drug Administration (FDA).
Hydroxyurea reportedly helps to increase Hb F production in patients, resulting in less pain and reduced hospital stays, according to the National Institutes of Health (NIH) National Library of Medicine journal.
It is set to be used in India, Africa and the Middle East.
Recently, a clinical study was conducted at the paediatric SCD Clinic of Korle Bu Teaching Hospital in Accra, Ghana.
In the study, the quantification results of Gazelle’s Hb F were within 4.5% of HPLC Hb F results, with a 95% confidence range.
Korle Bu Teaching Hospital’s Dr Catherine Segbefia said: “Part of the monitoring protocol for patients on hydroxyurea involves tracking changes in Hb F levels over time.
“Having this capability at the point of care with Gazelle’s Hb Variant test will allow greater access to this therapy, especially for those living in remote areas.”
Currently, the Hb Variant test is available in India as well as African and Middle East nations.





