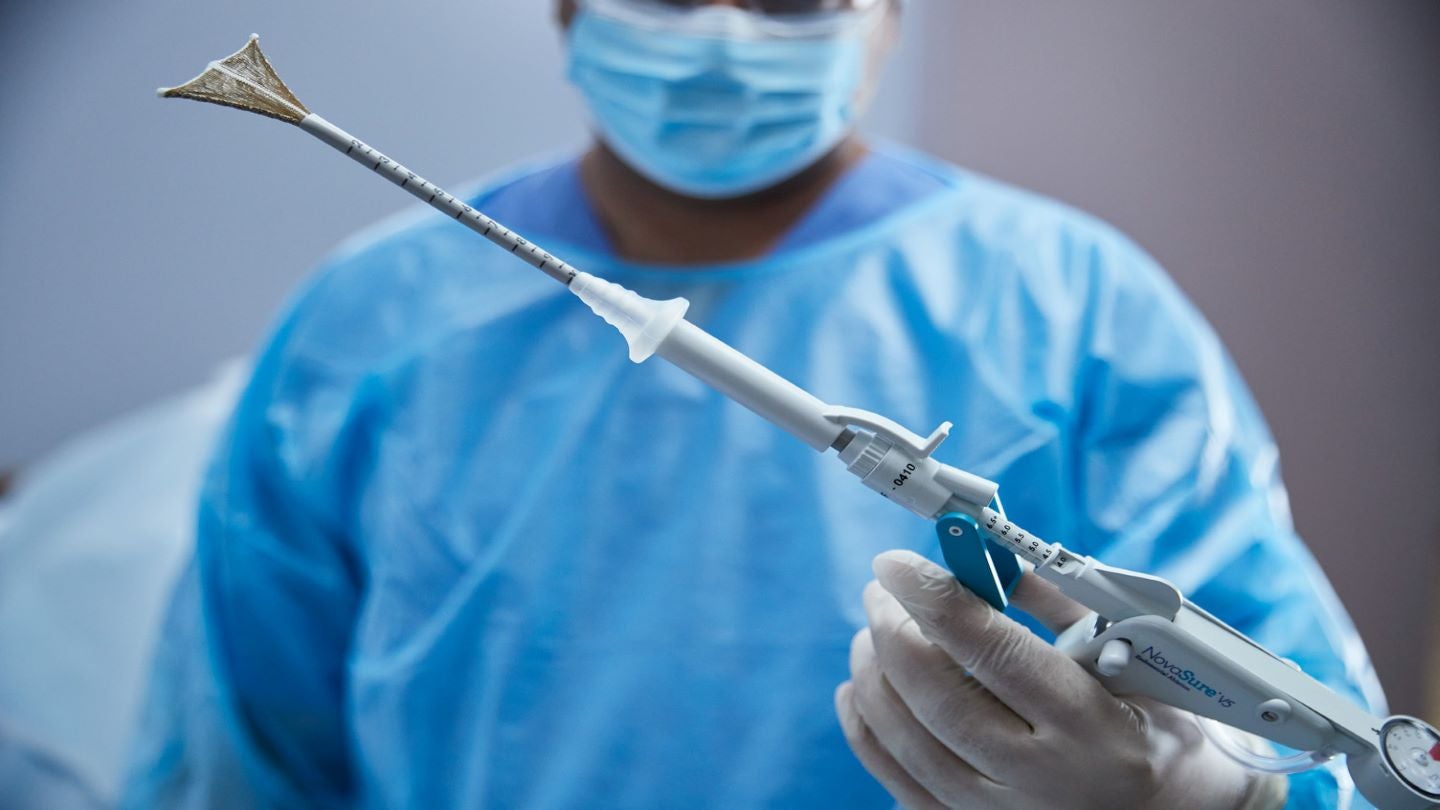
Hologic has received approval for its NovaSure V5 global endometrial ablation (GEA) device to be used in Canada and Europe.
The new version of the device has improved features to help treat several cervical and uterine anatomies.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
NovaSure V5 includes an updated cervical seal, which features EndoForm technology and is designed to increase the device’s sealing surface as well as accommodate a range of cervical canals and anatomies.
Its AccuSheath markings will enable improved accuracy and confidence for the seating and fundal placement, stated the company.
Furthermore, the device features SureClear technology, a fluid removal system that offers integrated suction through the array by constant tissue contact while eliminating ablation byproducts such as vapour and fluid.
In addition, the NovaSure device offers an endometrial ablation procedure to treat abnormal uterine bleeding.
Hologic international group president Jan Verstreken said: “Since its launch over 20 years ago, we have constantly listened to our customers’ feedback, which has driven continuous design improvements for NovaSure.
“This has enabled us to develop our technology to further meet their needs and those of the women they treat, ultimately delivering better outcomes for these women.”
Last July, Hologic launched the Panther Fusion SARS-CoV-2/Flu A/B/RSV assay and Novodiag RESP-4 molecular diagnostic test in the European Union (EU).
The assays were developed to detect and differentiate four respiratory viruses that can have similar clinical symptoms, namely influenza A (Flu A), influenza B (Flu B), SARS-CoV-2 and respiratory syncytial virus (RSV).





