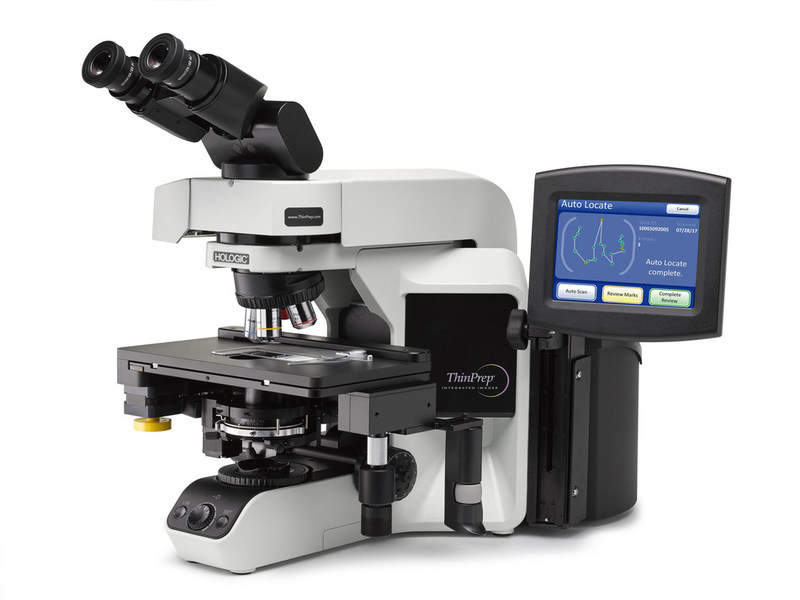
Hologic has secured premarket approval (PMA) from the US Food and Drug Administration (FDA) for its ThinPrep Integrated Imager designed for the automated imaging of Pap tests to detect cervical diseases.
The ThinPrep imager is a single, automated microscope that features a combination of Hologic’s ThinPrep computer-assisted imaging and dual slide review.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
With the capability to image slides within 90 seconds, the device allows cytology technicians to pinpoint areas that show potentially abnormal cells.
Previous studies indicated better sensitivity with automated, assisted imaging in identifying suspicious cells when compared to manual slide review.
Hologic Diagnostic Solutions division president Tom West said: “Approval of the ThinPrep Integrated Imager brings the benefits of ThinPrep automated Pap imaging to small and mid-sized laboratories in the US, and of course to their patients.
“Development of this newest ThinPrep product confirms Hologic’s commitment to women’s health and specifically to robust and accessible screening strategies for cervical cancer.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataThinPrep includes an imaging station and a review scope to ensure quality optics with an easy-to-use interface for a convenient, flexible workflow.
Along with the imager, Hologic is introducing its Compass Stainer in the US that can perform routine, as well as special staining protocols.
Last month, the FDA also granted PMA approval for the use of the diagnostics company’s Clarity 3D and Intelligent 2D imaging technologies on the 3Dimensions breast tomosynthesis system.
The system is designed to deliver higher resolution 3D images for radiologists, while offering comfortable, low-dose mammography experience to patients.





