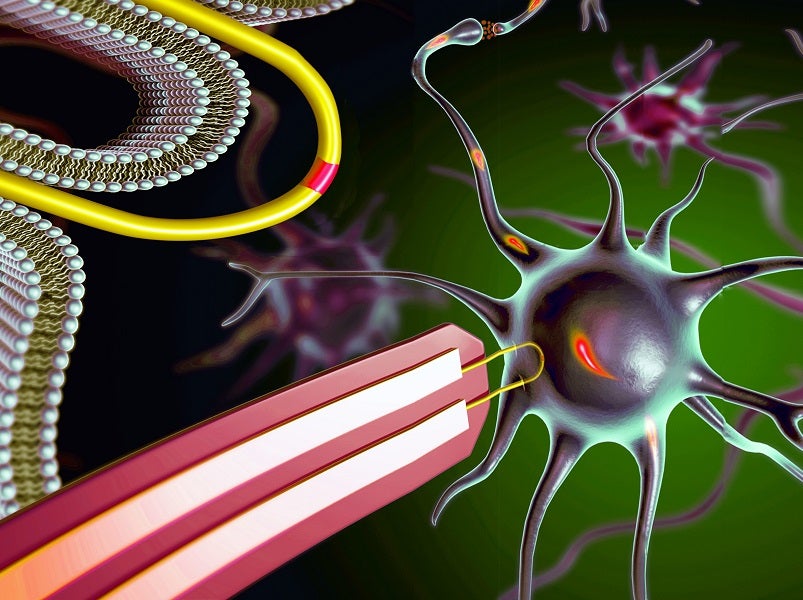
Researchers at the University of Surrey and Harvard University have manufactured scalable nanoprobe arrays small enough to record the inner workings of human cardiac cells and primary neurons.
The ability to read electrical activity from cells is the foundation of many biomedical procedures, including brain activity mapping and neural prosthetics. Developing minimally invasive new tools to read the electric current running within cells – known as intracellular electrophysiology – while still pushing the limits of what is physically possible has the potential to deepen scientists’ understanding of electrogenic cells and their networks in tissues.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This opens the door for new directions in human-machine interfaces.
Harvard University Department of Chemistry professor Charles Lieber said: “The beauty of science to many, ourselves included, is having such challenges to drive hypotheses and future work. In the longer term, we see these probe developments adding to our capabilities that ultimately drive advanced high-resolution brain-machine interfaces and perhaps eventually bringing cyborgs to reality.”
The research, published in the journal Nature Nanotechnology, details how the research team produced an array of the ultra-small U-shaped nanowire field-effect transistor probes for intracellular recording.
Current nanodevices with a similar function have struggled with a trade-off between device scalability and recording amplitude. The research team were able to address this by combining deterministic shape-controlled nanowire transfer with spatially defined semiconductor-to-metal transformation to realise scalable nanowire field-effect transistor probe arrays with controllable tip geometry and sensor size, which enabled the recording of up to 100 mV intracellular action potentials from primary neurons.
University of Surrey Advanced Technology Institute professor Yunlong Zhao said: “Our ultra-small, flexible, nanowire probes could be a very powerful tool as they can measure intracellular signals with amplitudes comparable with those measured with patch clamp techniques; with the advantage of the device being scalable, it causes less discomfort and no fatal damage to the cell. Through this work, we found clear evidence for how both size and curvature affect device internalisation and intracellular recording signal.”
The new device design allows for multiplexed recording from single cells and cell networks and could enable future investigations of dynamics in the brain and other tissues.





