Researchers at Massachusetts General Hospital (MGH) in the US have used magnetic resonance spectroscopy (MRS) imaging to investigate the mechanisms behind a potential targeted treatment for gliomas.
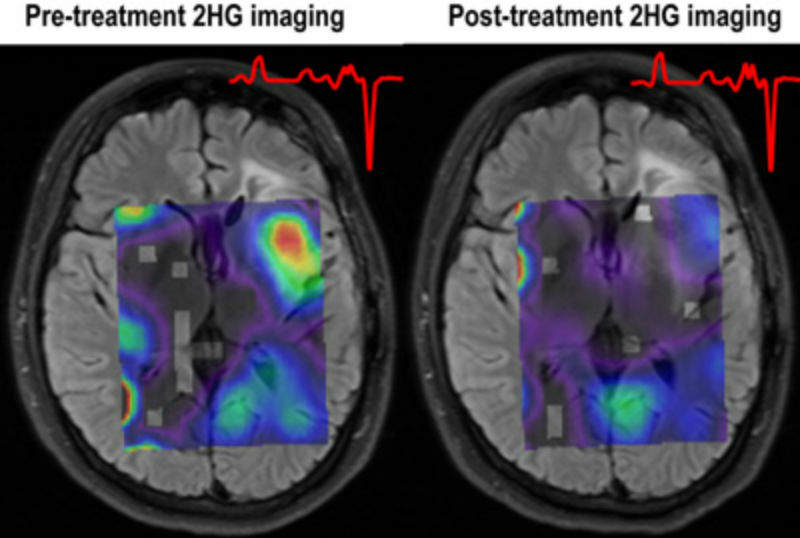
MRS imaging reflects metabolic rather than structural aspects of tissues, allowing the researchers to determine whether treatment with an investigational IDH1 inhibitor can reduce levels of a tumour-associated metabolite in patients with IDH1-mutated gliomas.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Harvard Medical School radiology assistant professor and lead author of the study Dr Ovidiu Andronesi said: “Gliomas are aggressive, primary brain tumours that lack effective treatments, and patients invariably succumb to the disease. There is a desperate need for progress in glioma treatment, and IDH mutations, which occur commonly in these tumours, offer a pathway for targeted therapy.
“With the new metabolic imaging method that we developed to probe treatment effects, we showed that a novel IDH1 inhibitor can quickly reduce levels of the oncometabolite 2HG in patients with this mutation. This type of methodology has the potential to accelerate clinical trials and translation of targeted therapies, such as mutant IDH inhibitors, and makes the concept of precision oncology feasible in glioma patients.”
Patients with gliomas characterised by mutations in the isocitrate dehydrogenase (IDH) enzyme live three to five times longer on average, with better response to chemo and radiation therapy than patients with tumours that do not have IDH mutations. However, the mutations themselves may initiate and drive tumour growth.
IDH mutant tumours produce higher levels of 2-hydroxyglurate (2HG), believed to contribute to tumour initiation by interfering with gene expression control. It is unclear whether reducing 2HG levels could reverse the process but it could be used as a diagnostic biomarker for the monitoring of IDH mutant tumours.
Of eight patients who had enrolled in the trial at the time of the current study, MR imaging data collected before and a week after treatment began was available for five. 3D MRS imaging of this data found that 2HG levels dropped around 70% after treatment initiation. The levels of additional metabolites connected with the mutant IDH1 pathway were altered in response to IDH305 treatment. This indicates the potential metabolic reprogramming of the tumour.
Andronesi added: “MR spectroscopy is quick, non-invasive and can be performed on any clinical MR scanner, making results available before any biopsy or surgical procedure have begun.
“It also can be used to track the response to treatment, since repeat biopsies are not feasible for patients with brain tumours, by directly probing the activity of the mutant IDH1 enzyme. Of course, our small study needs to be replicated in a larger group with longer follow-up times, as well as determining the long-term benefit of IDH1 inhibitor treatment.”





