Researchers at MIT have collaborated with Beta-O2 Technologies to develop an implantable device that can provide transplanted islet cells with a supply of oxygen so they have a chance of treating type 1 diabetes.
As islet cells are responsible for producing insulin when blood glucose concentrations increase, researchers have been attempting to find a way that an islet transplant could treat type 1 diabetes for decades.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The main challenge is that once islets are transplanted they die if they do not receive an adequate supply of oxygen. The new implantable device overcomes this problem as it contains an oxygen supply centre for the islet cells that can be replenished every 24 hours.
MIT professor of chemical engineering and senior author of the study Clark Colton said: “Getting oxygen to these cells is a difficult problem.
“The benefits of this approach are: you keep the islets alive to perform their function, you don’t need as much tissue, and you reduce the ability of the implants to provoke an immune response.”
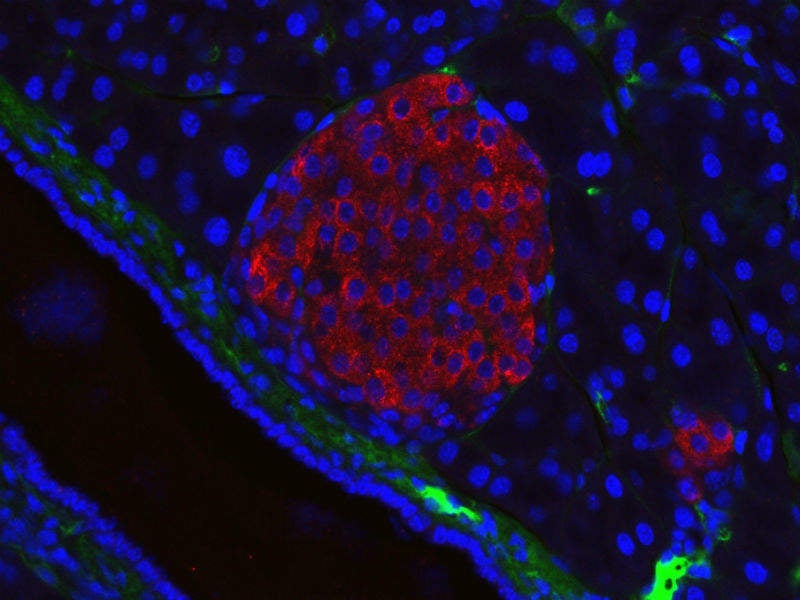
Islet cells in a healthy pancreas receive oxygen-rich blood from capillaries but when surgeons first tried to transplant islets into diabetic patients, many of the cells did not have direct contact with capillaries.
Colton’s previous research found that the outer surface of islets needs to be exposed to at least 50 mm Hg of oxygen to remain viable and produce insulin normally. The researchers then determined the operating conditions of the device needed for islets to function for long periods of time while assembled in a compact form small enough to be implanted in human patients.
The implant device they designed encapsulates islets in a slab of alginate, a material produced by algae, around 600 microns thick. The slab has a membrane on one side that protects it from immune cells and large proteins but allows insulin, nutrients and oxygen through. Below this layer is the gas chamber, which is about 5mm thick and contains gases such as nitrogen, carbon dioxide and oxygen. Oxygen flows from the chamber through the membrane and into the islets embedded in the alginate slab.
Oxygen diffusing through the slab is gradually consumed so the oxygen partial pressure continually drops. To ensure that this pressure remains at least 50 mm Hg for 24 hours, it was found that a 500 mm Hg oxygen partial pressure was needed in the gas chamber to begin with.
After 24 hours, the oxygen supply is replenished through a port which sits under the skin and is connected to a catheter that leads to the encapsulated islets.
When the implants were tested in a rat model, 90% of the islets remained viable for several months and most rats maintained normal blood glucose levels throughout that time. However, they returned to diabetes levels after the implants were removed.
Another benefit of the approach is that a patient’s immune response to the transplant is minimised because most of the islet cells remain alive and therefore do not break down and attract the attention of the immune system.
Beta-O2 Technologies researchers are now developing new versions of the device which feature an oxygen storage chamber that can be implanted below the skin but separate from the islets. This version would only need to be replenished once a week.





