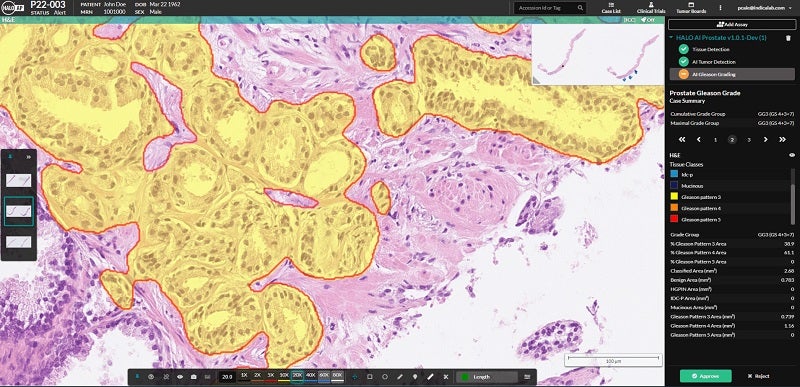
Indica Labs has received CE-IVD Mark for its screening tool, HALO Prostate AI, which will enable pathologists to identify and grade prostate cancer in core needle biopsies.
Working alongside pathologists, the deep learning-based tool will add a quality control layer for accurate diagnosis as well as improve efficiency.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
HALO Prostate AI has been developed in partnership with Dr Yuri Tolkach and colleagues from the University of Cologne, Germany.
Clinically validated against the Hamamatsu NanoZoomer S360 and Leica GT450 platforms, the tool was trained using more than 870,000 training patches.
These patches were obtained from the annotation of digital scans that represent the complete range of prostate cancer subtypes and Gleason grades, in addition to benign tissue.
In validation studies, the algorithm achieved 95% to 100% sensitivity, 88% to 98% specificity and a negative predictive value of 98% to 100%.
The study was conducted on 4,973 core needle biopsies obtained from three independent arms from Austrian and German hospitals.
In another study, Gleason scores obtained from the tool were compared against scores that were assigned by pathologists in ten hospitals.
Dr Tolkach said: “With tools such as HALO Prostate AI, our work can be substantially optimised while controlling for high-quality, reliable and objective diagnostics.
“HALO Prostate AI showed very high accuracy in the large multi-institutional study for tumour detection and Gleason Grading in prostate biopsies.”
Deployed through the company’s CE-IVD marked HALO AP platform, the screening tool will provide an end-to-end workflow that is fully validated and automated.
HALO Prostate AI is also designed to be scanner agnostic.
Indica Labs AI Collaboration principal scientist Dr Peter Caie said: “We set out to build a clinical-grade algorithm that could improve turn-around time and diagnostic accuracy for prostate cancer patients, and I believe that is exactly what we have achieved.”





