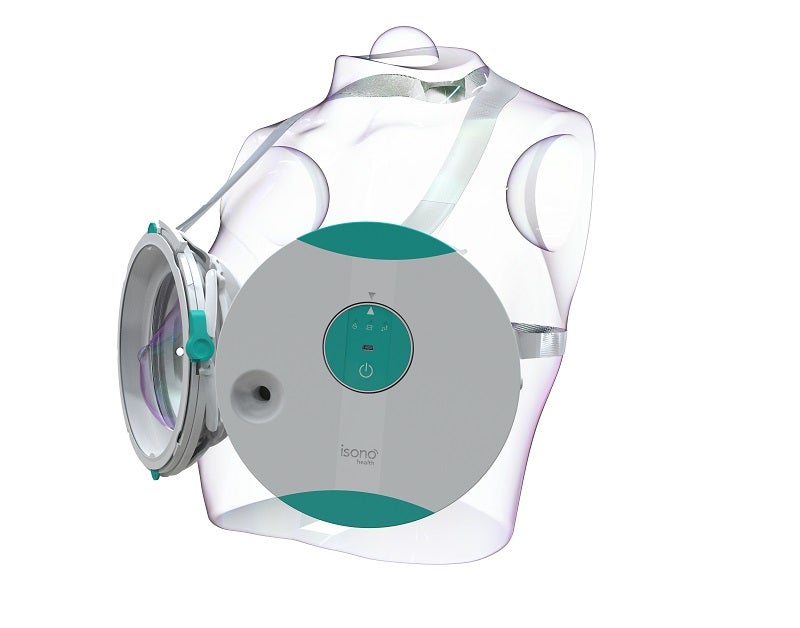
iSono Health has received approval from the US Food and Drug Administration (FDA) for its ultrasound wearable system, which has been designed for personalised breast imaging.
Said to be the world’s first compact automated wearable 3D breast ultrasound, the ATUSA System features intuitive software for automated image acquisition and analysis.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The wearable accessory and intuitive user interface of ATUSA ensure easy, quick and comfortable image acquisition.
iSono Health co-founder and CEO Maryam Ziaei said: “Breast cancer is the leading cause of cancer death among women worldwide, and one in eight women will be diagnosed with breast cancer in their lifetime.
“We founded iSono Health with the vision to enable earlier diagnosis and treatment for breast cancer to save women’s lives, and this FDA clearance is a major step to fulfilling that vision.”
The patented technology of ATUSA is designed to provide accuracy, improved efficiency and a comfortable experience for patients.
Additionally, patients and physicians can access 3D breast ultrasound imaging at point of care.
ATUSA scans the complete breast volume automatically, provides 3D visualisation of the tissue in just two minutes and does not need a trained ultrasound operator.
The scanner will also integrate with the latest machine learning models, giving physicians a complete toolset for decision making and patient management.
iSono Health is currently carrying out prospective case collection studies for further validation of various deep learning models integrated with ATUSA.
This will aid clinicians in breast lesions localisation and classification.
Ziaei further added: “Clinicians and women worldwide need high-quality breast imaging that is accessible and efficient at scale, without the need for highly skilled operators.
“The portable and automated ATUSA system stands alone in comparison to other ultrasound offerings in promising to address that need.”





