iSTAR Medical has received investigational device exemption (IDE) from the US Food and Drug Administration (FDA) to commence a clinical trial of its minimally invasive implant for glaucoma surgery (MIGS), MINIject.
MINIject is intended for treating patients suffering from primary open-angle glaucoma.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
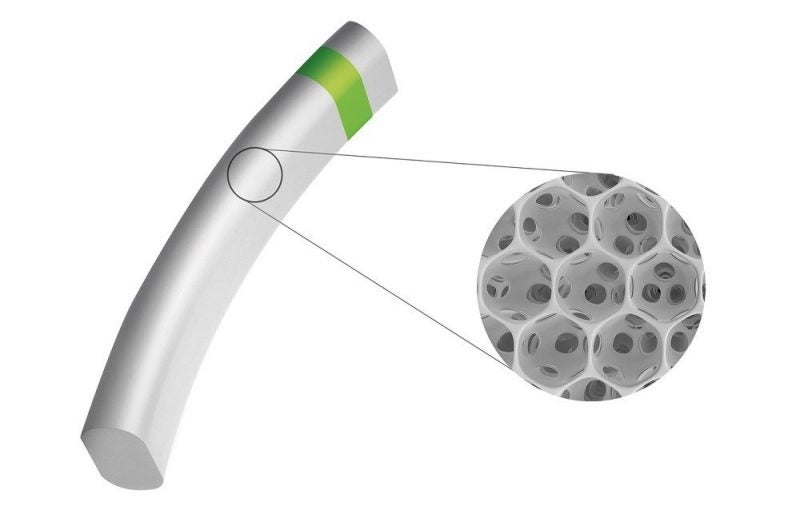
The device integrates the porous structure of the company’s STAR material with the power of the supraciliary space.
It can boost natural fluid outflow and lower intraocular pressure (IOP) and medication requirements, as well as bio-integrate with the adjacent tissue and reduce inflammation, fibrosis and future complications.
The STAR-V trial will analyse the efficacy of MINIject by determining the mean decrease in eye pressure and proportion of subjects that experience a minimum of 20% reduction in eye pressure.
It will assess the safety and efficacy of only the device in a procedure that does not merge with concurrent cataract surgery.
The trial will enrol more than 350 primary open-angle glaucoma patients with glaucoma surgeons in the US, Canada and Europe taking part.
Participants will be followed up to analyse MINIject’s long-term benefits and tolerability for treating mild to moderate glaucoma.
The important findings from the trial will be made available after all subjects complete two years in the study, the company noted.
iSTAR Medical CEO Michel Vanbrabant said: “We are very pleased that the FDA has granted us approval to bring this innovative technology to North American patients suffering from primary open-angle glaucoma in the STAR-V trial.
“Results from clinical trials in over 130 patients in Europe, Asia and Latin America have consistently demonstrated that MINIject maintains a positive safety profile, and delivers a significant reduction of pressure thanks to our proprietary STAR material and the power of the supraciliary space.”
In September 2019, iSTAR Medical raised €40m in a Series C funding round to develop new implants for the treatment of glaucoma.





