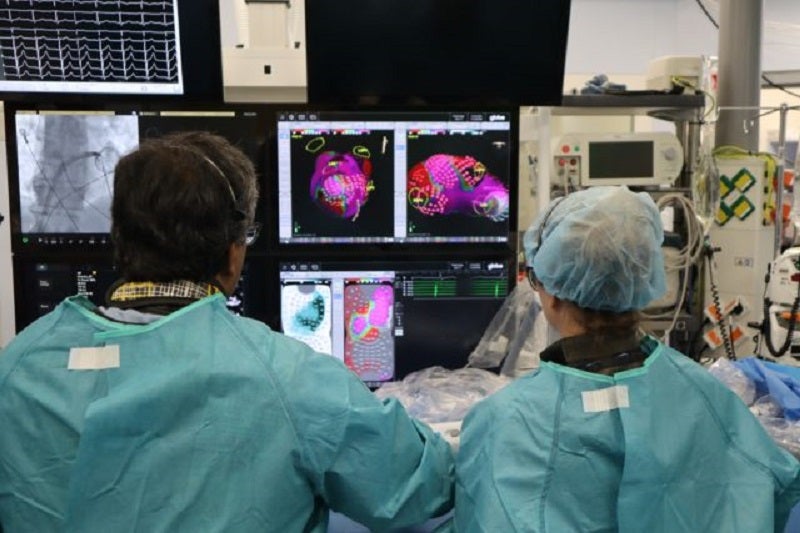
Canadian medical solutions company Kardium has successfully completed the first-in-human clinical study of its Globe Pulsed Field (PF) System for treating atrial fibrillation (AF) using pulsed field ablation (PFA) therapy.
The paroxysmal AF patients in the study received pulmonary vein isolation (PVI) treatment, which took roughly 16 minutes.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
After this, the next-generation Globe PF System was used to create a high-definition map of the complete atrium using real-time 3D mapping capabilities.
Persistent AF patients received additional ablation of the posterior wall, and many of them also received a mitral isthmus line using the same Globe Catheter.
The Kardium team worked with Dr Vivek Reddy from Mount Sinai Hospital in New York, US, as well as professor Petr Neužil and Dr Jan Petrů from Na Homolce Hospital in Prague, Czech Republic, to treat 38 patients.
They used the Globe PF System to isolate 100% of the patients’ pulmonary veins.
Kardium CEO Kevin Chaplin said: “The Globe PF System is designed to be a safe, effective and rapid treatment for AF.
“We are extremely excited by these procedures, which demonstrate the flexibility and ease of use of the Globe PF System.
“Previous clinical cases with the Globe PF System have achieved 100% durable PV isolation three months after the procedure.”
The Globe PF System comprises a Globe Catheter with 122 electrodes. Each electrode can map the patient’s cardiac anatomy as well as electrical activity.
They can also deliver pulsed field ablation energy to the heart for the treatment of AF.
The Globe Catheter determines the electrodes that are in contact with cardiac tissue using contact sensing to ensure therapy is provided effectively to the patient’s heart.
Furthermore, the company intends to conduct an international pivotal clinical study of the Globe PF System in Germany, Canada, the Czech Republic and the US to obtain regulatory approval for its commercial sales.





