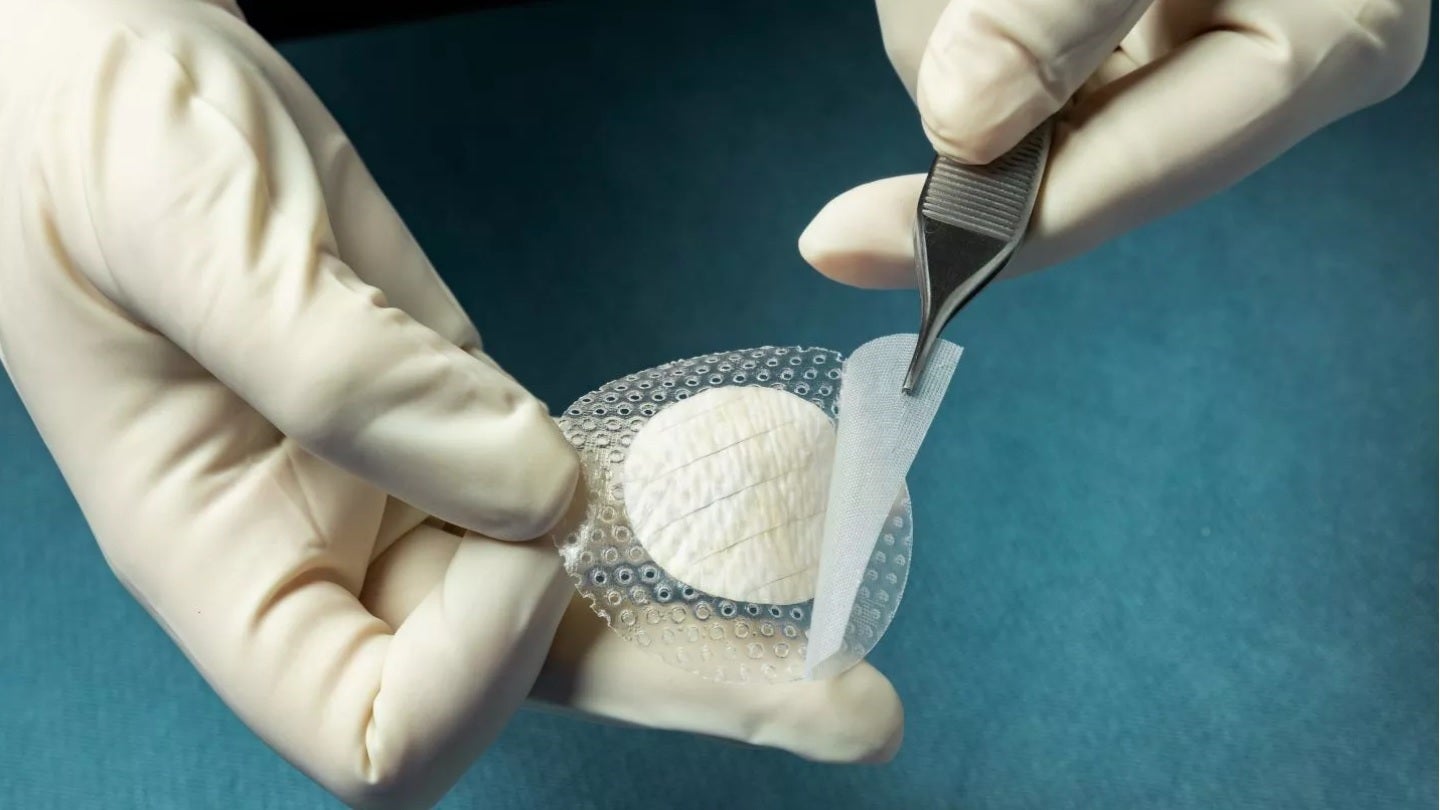
Medical fish skin company Kerecis has launched the MariGen Shield – a solution that combines the company’s fish-skin graft and a silicone contact layer to treat chronic and complex wounds.
The latest product is fenestrated to enable proper drainage of wounds. It creates an ideal environment for wound management and ensures optimal moisture levels that promote effective healing.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The integrated, overlapping silicone contact layer anchors the round graft in position, helping to safeguard the granulating wound and the fish-skin graft following the application of a protective cover dressing.
Kerecis is offering MariGen Shield in round grafts of 20mm and 30mm for application to foot wounds.
Kerecis founder and CEO Fertram Sigurjonsson said: “Until now, the application of the Kerecis fish-skin graft has required three components: the fish skin itself, a contact layer and a cover dressing to regulate the moisture level on the wound.”
“By integrating the contact layer with the fish skin, the application becomes faster and easier and the inventory needs at the healthcare facility simpler.”
Additionally, the company has disclosed the findings of a clinical trial that evaluated the efficacy of Kerecis fish-skin grafts in comparison to a standard of care for the management of diabetic foot ulcers.
The findings along with the new product were announced at the Symposium on Advanced Wound Care event in Maryland, US.
The multicentre and comparative study demonstrated that the MariGen outperformed Fibracol therapy by nearly doubling the healing rate of non-responsive, chronic diabetic foot ulcers.
The trial was carried out across 16 centres in the US, involving a total of 102 patients between June 2019 and January last year.





