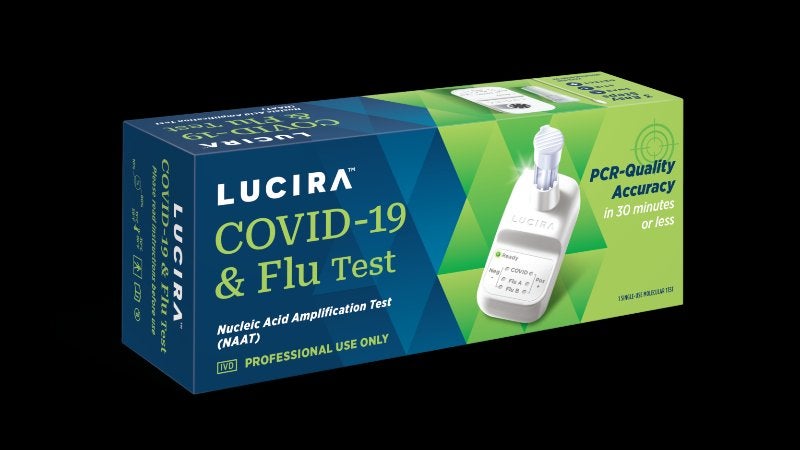
Medical technology company Lucira Health has obtained CE mark for professional use for its Covid-19 & Flu and Covid-19 molecular tests.
The marking will enable the company to sell and distribute the two tests throughout the European Union (EU).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The two single-use Nucleic Acid Amplification Tests (NAAT) are miniaturised, to fit in the palm, and offer results in 30 minutes.
The Lucira Covid-19 test uses genetic material from a swab to detect the virus. The test includes a test device and two AA batteries, as well as a sample vial, swab and information leaflet. It does not require any other instruments to provide the test results.
Currently, the test is available in the US, Canada, Singapore, Israel and Taiwan.
The other test, Lucira Covid-19 & Flu test, can diagnose Covid-19, Flu A and Flu B. It also runs on two AA batteries.
The company plans to roll out the Covid-19 & Flu test in the EU from the start of the year’s third quarter, before the influenza season.
Lucira Health president and CEO Erik Engelson said: “The Lucira testing platform can make highly accurate molecular point-of-care testing more accessible than previously possible with our simple test that requires no separate instrument or reader, internet connection, or power cord.
“Receiving CE Mark for both our Covid-19 & Flu and Covid-19 tests represents a major milestone for Lucira to expand our product line and our geographic reach.”
In March, Baebies obtained CE Mark for its Finder SARS-CoV-2 point-of-care test.





