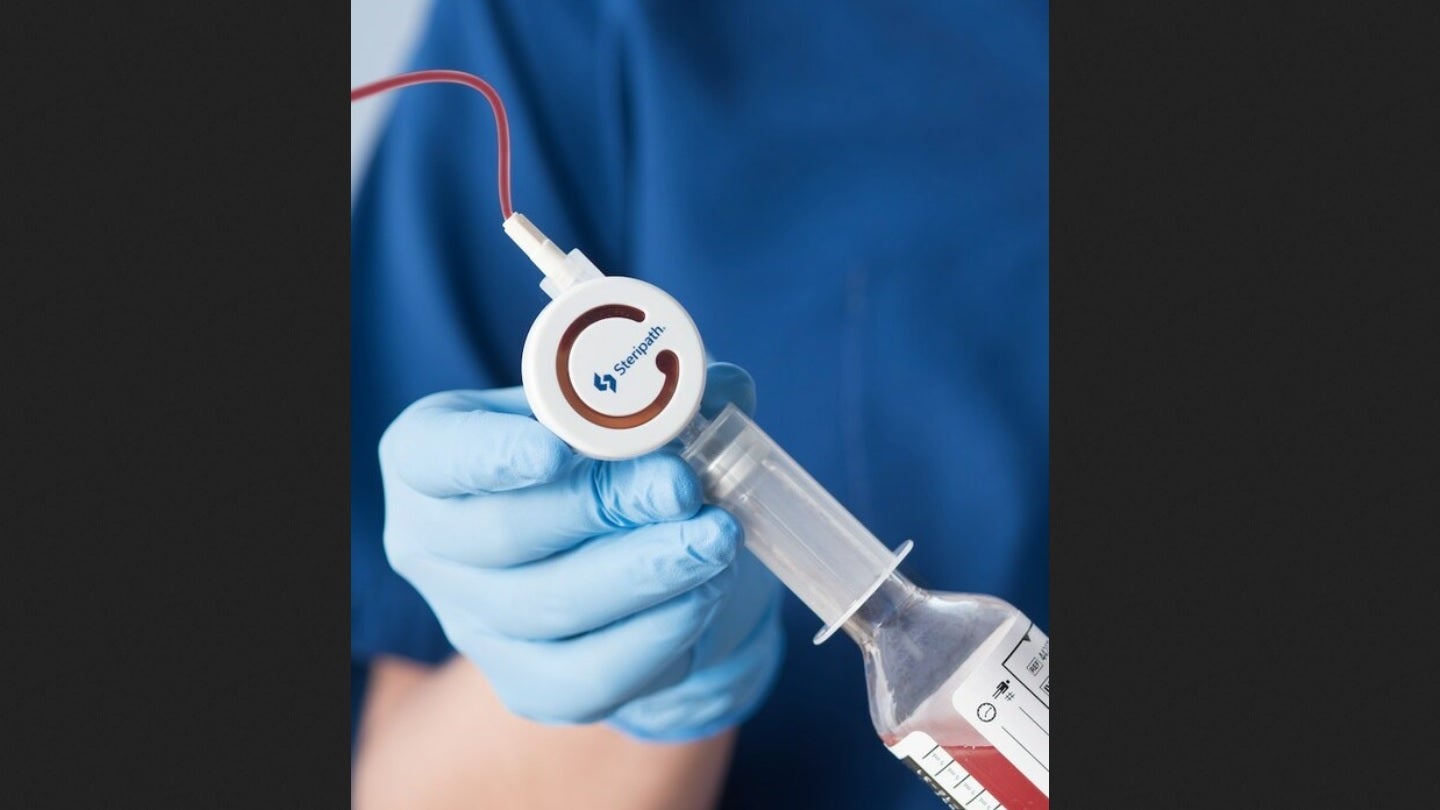
Magnolia Medical Technologies has obtained 510(k) clearance from the US Food and Drug Administration (FDA) for 19 new Steripath Micro configurations within its Initial Specimen Diversion Device (ISSD) product family.
Hospitals can use the new options including direct-to-bottle and BD VacutainerUltraTouch push-button blood collection set configurations, to meet the requirements of all patient populations.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Magnolia Medical CEO Greg Bullington said: “We are delighted to launch the expanded family of Steripath Micro configurations as an integral part of our Initial Specimen Diversion Device portfolio.
“We developed the Steripath Micro platform in close collaboration with our customers to ensure the ability to provide improved blood culture accuracy for all patient populations including those that are most vulnerable.”
Steripath Micro is claimed to be the only FDA 510(k)-approved low-diversion volume blood culture collection device family with a specific indication for the reduction of blood culture contamination.
Magnolia Medical developed Steripath Micro in partnership with several hospital-based clinicians. The system optimises the blood culture collection process.
All needle configurations in Steripath Micro incorporate the BD VacutainerUltraTouch push-button blood collection set.
UltraTouch needles feature an ultra-thin–wall cannula, which extends the inner diameter while maintaining a true–to–gauge size outer diameter.
With the BD PentaPoint cannula technology incorporated, the BD VacutainerUltraTouch is able to easily penetrate the skin for a comfortable patient experience.
In October last year, Becton, Dickinson and Company (BD) signed a co-exclusive commercial agreement with Magnolia Medical to co-sell and co-market the latter’s Steripathand SteripathMicro ISDDplatforms.





