Magnus Medical has secured US Food and Drug Administration (FDA) 510(k) clearance for its SAINT Neuromodulation System to treat major depressive disorder (MDD).
The individualised, non-invasive system has been designed to treat MDD in adult patients who have not achieved sufficient recovery after receiving antidepressant medication in the current episode.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The SAINT system is said to be an innovation that will significantly impact the treatment of severe depression.
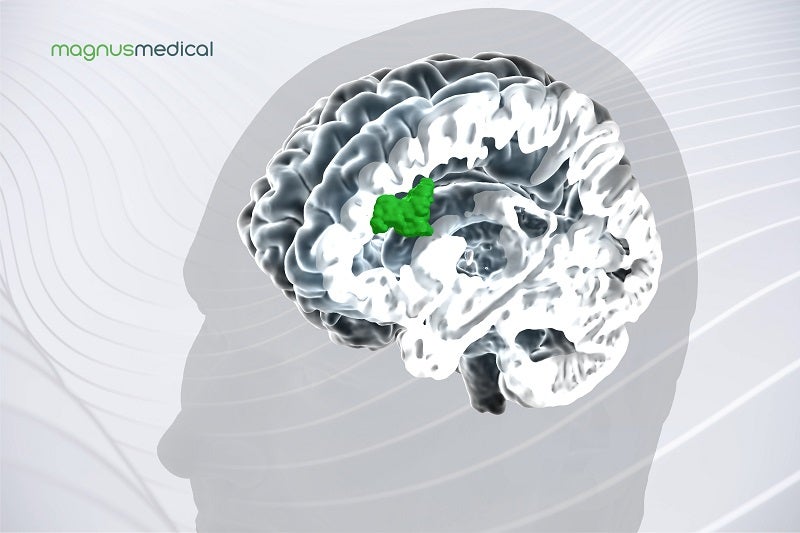
It combines advanced imaging technologies, personalised targeting and new stimulation patterns.
According to Magnus Medical, this combination yields a new form of individualised neurostimulation for people with treatment-resistant depression.
Using structural and functional magnetic resonance imaging (MRI), the system informs a proprietary algorithm.
The algorithm identifies the optimal anatomic target for focused neurostimulation in MDD patients.
Last October, the SAINT Neuromodulation System received Breakthrough Device Designation from the US FDA.
Findings from a double-blinded randomised controlled trial (RCT), published in the American Journal of Psychiatry, demonstrated that 79% of the people who received active treatment with the system have entered remission from their depression.
Magnus Medical co-founder and CEO Brett Wingeier said: “We are now at the forefront of an enormous improvement in the care of treatment-resistant depression, thanks to the work of the Magnus team and all those whose efforts have led to the SAINT technology.
“Today’s FDA’s clearance for the SAINT Neuromodulation System is a major milestone in our long-term journey to restore and sustain mental health.
“Our clinical research programme will continue to yield more insights into how personalised neuromodulation can restore healthy neural activity across a wide variety of mental health conditions.”
Wingeier further added that the company expects to commercially launch its Neuromodulation System on a limited basis later next year.





