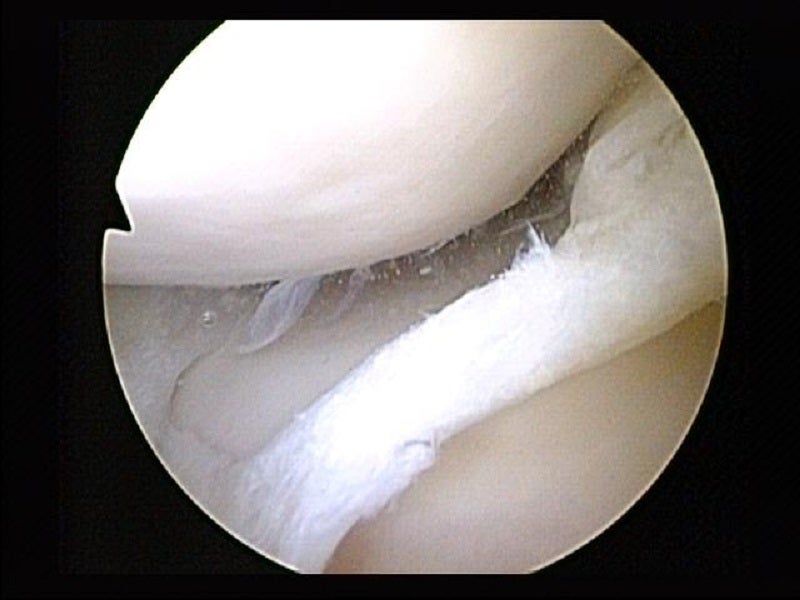
The Marshall University Joan C Edwards School of Medicine in West Virginia, US, has started accepting applicants in order to launch a clinical trial of a new technology for performing meniscus repairs.
Set to launch this year, the prospective randomised trial has been designed for comparing nanoscopic and standard arthroscopies for meniscus tears treatments.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Its aim is to determine whether the minimally invasive nanoscope approach improves patient outcomes for partial meniscectomies.
The trial will also include a complete assessment of the patient’s return to work and play following the surgery.
Marshall University Joan C Edwards School of Medicine orthopaedic surgery assistant professor Chad Lavender said: “We have developed several nanoscopic techniques at Marshall and have been pleased with the minimally invasive nature and positive impact on our patients. This trial is the next step in that research.”
Currently, the university is recruiting 100 participants aged under 60 years with a meniscus tear for the trial.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataIn the study, the participants will be randomly divided into two equal groups. One group will receive surgery using nanoscopic technology, while the other will be treated using standard arthroscopy.
Marshall University stated that the surgeries will be performed at Cabell Huntington Hospital in Huntington, West Virginia.
For the ongoing assessment, all study participants will regularly attend the scheduled study visits with Marshall Health physicians.
Nanoscope distributor, Arthrex has provided partial funding for the prospective randomised trial.
Physicians use a needle scope, which is called a nanoscope, instead of standard portals and incisions to perform a nanoarthroscopy.





