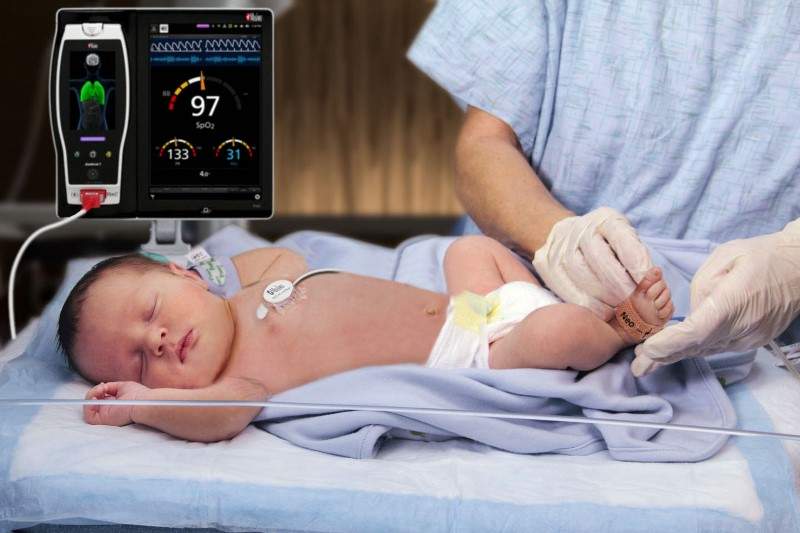
Medical technology company Masimo has received FDA clearance for its Rainbow Acoustic Monitoring (RAM) RAS-45 acoustic respiration sensor for infant and neonatal use.
Earlier, the company’s RAS-125c and RAS-45 sensors were cleared to monitor adult and paediatric patients weighing more than 10 kg.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The latest clearance makes acoustic respiration rate measurement possible in patients of all age groups including neonates in the US.
The RAS-45 sensor expands the continuous respiration rate (RRa) accuracy range to 120 breaths per minute as well as offers accuracy of +/- 1 breath per minute.
The sensor is smaller than the RAS-125c sensor and is suitable for the smaller stature and delicate skin of infants and neonates.
Masimo founder and CEO Joe Kiani said: “From the beginning, we have focused our R&D on neonates and children for many reasons, including our belief that helping clinicians care for children will provide more benefit to society.
“RAM harnesses the power of our breakthrough signal processing and sensor technology and applies it to a measurement that has either been unreliable or difficult to use, respiration measurement, the third vital sign.”
According to the company, RAM measures respiration rate by applying an adhesive sensor with an integrated acoustic sensor, such as RAS-45 and RAS-125c, to the patient’s neck area.
Leveraging the acoustic sensors, clinicians can listen to the sound of a patient’s breathing through a point-of-care device such as Radical-7 Pulse CO-Oximeter, or from a Patient SafetyNet view station.





