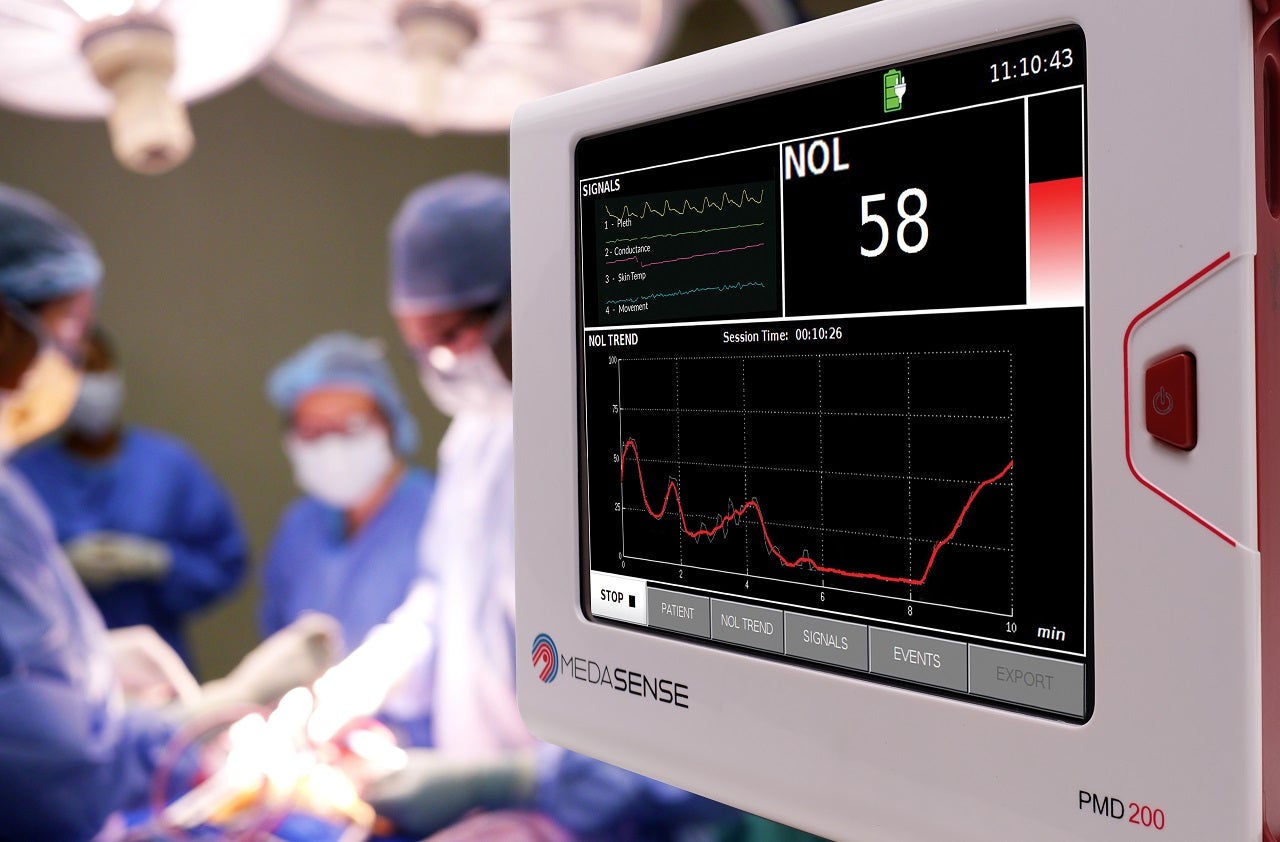
Israel-based Medasense Biometrics has received approval to market its NOL pain response monitoring device (PMD-200) in Brazil.
The device is used by surgical teams to manage pain medication during surgery when the patient is under anaesthesia and unable to communicate.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Medasense’s NOL technology uses a non-invasive sensing platform and an artificial intelligence (AI) algorithm to objectively monitor and quantify the individual patient’s pain response.
It helps clinicians to optimise and personalise pain control and eliminate the chance of overmedication.
NOL technology is currently being assessed in the intensive care unit for mechanically ventilated patients, including Covid-19 patients, the company noted.
The outbreak of the Covid-19 pandemic in Brazil has resulted in a spike in the number of mechanically ventilated patients, requiring skilled pain management and optimal analgesia to help improve recovery.
Medasense has partnered with Brazilian company JG Moriya for the commercial launch of NOL pain response monitoring device in Brazil.
JG Moriya CEO Karine Moriya said: “Our network of influential surgeons and anesthesiologists should allow us to bring this much-needed technology to Brazil’s advanced surgical facilities and we expect to perform the first NOL-guided surgeries in the coming weeks.”
In September, Medasense Biometrics raised $18m in a Series C round from Sabadell Asabys venture capital company, Israeli family offices, and returning investors Baxter Ventures, Olive Tree Ventures, and LGL Capital.
The company will use the funds to expand and further consolidate commercial deployment of NOL technology in Europe through the distribution agreement with Medtronic.
Medasense is also pursuing the Food and Drug Administration (FDA) approval of the NOL pain response monitoring device in the US.





