Median Technologies has signed a research collaboration agreement with the University of California San Diego in the US (UC San Diego) to conduct a study for validating the former’s proprietary imaging platform, iBiopsy.
Based on artificial intelligence (AI) technology, iBiopsy targets developing non-invasive image-based diagnosis tests and solutions for use in indications that have unmet needs.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Currently, various indications are targeted for liver diseases and the use of immuno-oncology drugs.
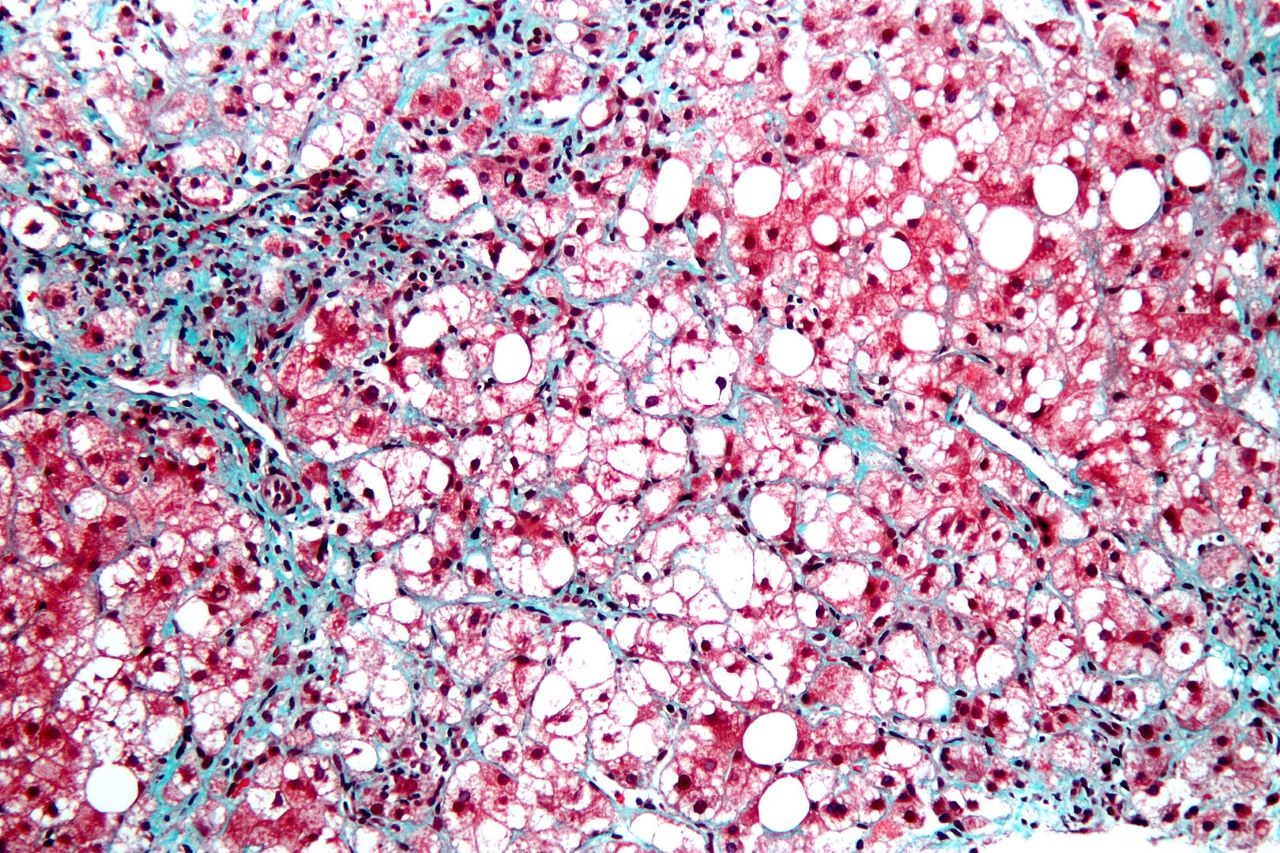
The study on liver fibrosis severity in Non-Alcoholic Steato-Hepatitis (NASH) patients is linked to the iBiopsy NASH Clinical Development Plan (CDP).
Estimates show that NASH affects 1.5%-6.45% of the global population and no cure for the advanced stage of the disease exists.
The clinical aim of Median’s retrospective study with UC San Diego is to quantify iBiopsy’s ability in learning algorithms to distinguish between early and advanced fibrosis grade in NASH patients.
UC San Diego Radiology professor, diagnostic radiologist and study’s principal investigator Dr Kathryn Fowler said: “Magnetic Resonance Imaging is well-positioned to provide biomarkers for non-invasive diagnosis and longitudinal monitoring in patients with fatty liver disease.
“The development and validation of these biomarkers is a primary focus of the Liver Imaging Group at the University of California, San Diego.”
The study will be carried out retrospectively on a cohort of over 300 patients and the company anticipates confirming the previously reported results on the NASH Clinical Development Plan.
First results from the Median-UC San Diego study are expected in the first quarter of next year.
Furthermore, the partnership will aid in advancing the clinical research intended to improve NASH patient diagnosis and monitoring.
Median Technologies founder and CEO Fredrik Brag said: “The development of a non-invasive biomarker to diagnose early NASH is critical for patients.
“This major collaboration will complement our first validation steps in 2020 and support our go-to-market strategy.”





