UK-based medical device start-up Medovate has obtained the exclusive licence to design, develop, manufacture and launch two complementary local anaesthesia products.
One of the products, Regional Anaesthesia Device (RADS), has been created in alliance with Leeds Teaching Hospitals NHS Trust. It is a portable device that requires only one operator.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
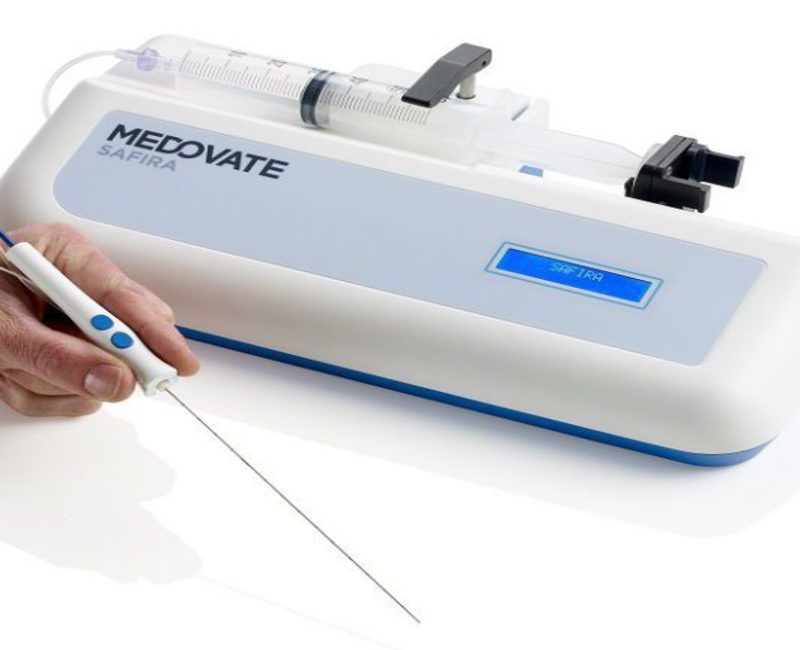
Medovate also secured rights to Safer Injection for Regional Anaesthesia (SAFIRA), which is being developed with the Queen Elizabeth Hospital, King’s Lynn, NHS Foundation Trust.
SAFIRA is designed to offer end-to-end safety via a simple and quick method to inject regional anaesthetic agents. It allows enhanced data monitoring by recording the whole procedure with specific readings on time, pressure and volume.
Both RADS and SAFIRA are intended to deliver regional blocks within safe pressure limits.
Under the terms of the licensing agreements, Medovate will be responsible for the design and development, regulatory approval, and the clinical trial to market launch of the products.
Medovate managing director Stuart Thomson said: “There are approximately six million regional block procedures carried out in the EU and US each year. Injection above the published safe pressure threshold can lead to serious nerve damage in up to 1% of cases and transient nerve damage in up to 5% cases, so this is a major problem.
“There are also broader repercussions including financial compensation claims, increased anaesthetist and care provider anxiety and importantly, a rise in patient re-admission. RADS and SAFIRA together can tackle all these problems and help ensure that complications from nerve damage finally become a thing of the past.”
Launched by Health Enterprise East (HEE) in February this year, Medovate focuses on the development and commercialisation of medical technologies.
The company aims to offer financial and technical support to med tech innovators within the NHS.





