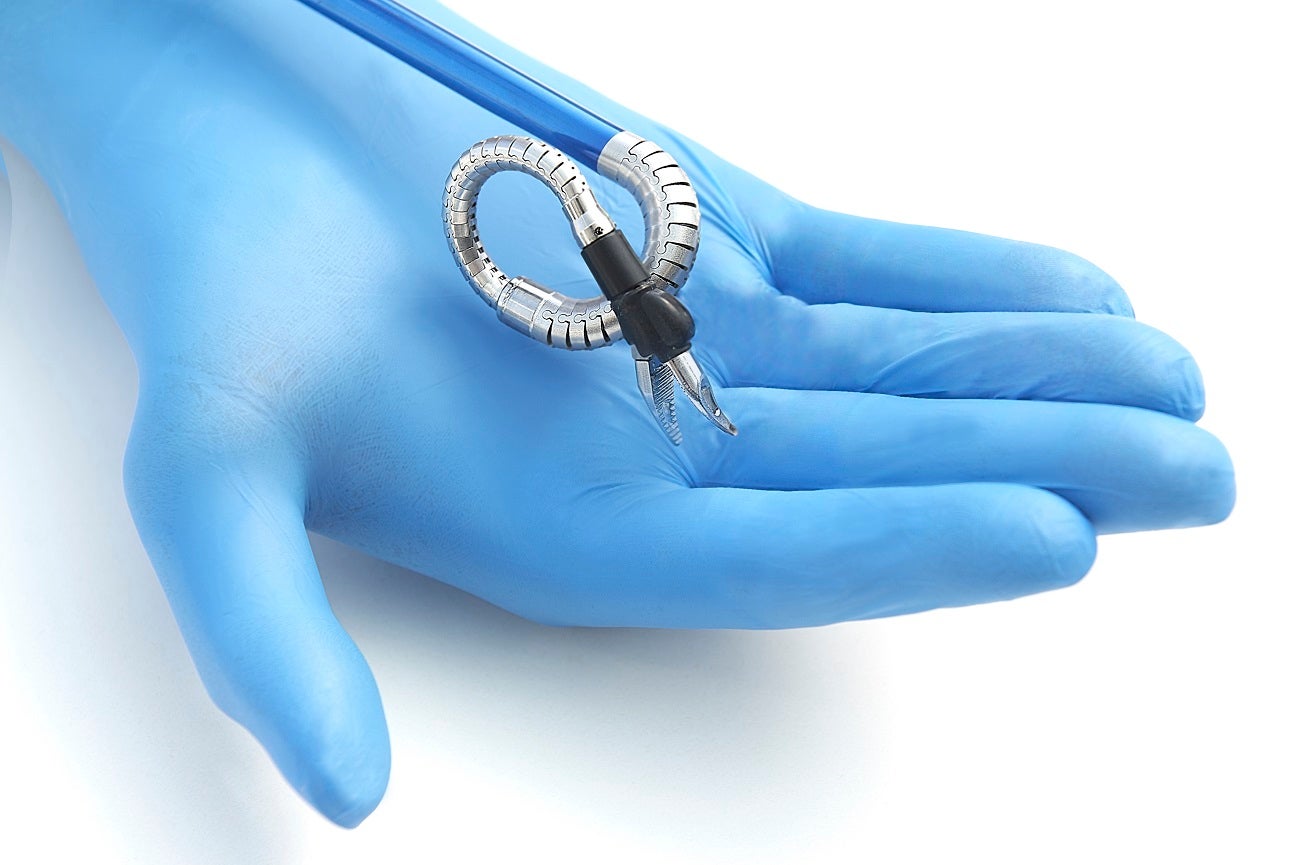
The US Food and Drug Administration (FDA) has granted De Novo authorisation to the first ever robotically-assisted surgical device for benign transvaginal surgical procedures.
The Hominis surgical robotics platform, developed by Israeli robotically-assisted surgery company Memic Innovative Surgery, is now approved for use in single-site, natural-orifice laparoscopic-assisted transvaginal benign surgical procedures.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
This includes benign hysterectomy (removal of the uterus for non-cancerous conditions) with salpingo-oophorectomy (removal of one or both fallopian tubes and ovaries).
The device uses surgical instruments that are inserted through the vagina and a video camera inserted laparoscopically through a single abdominal incision, making Hominis procedures far less invasive than traditional laparoscopic hysterectomy.
The device features humanoid robotic arms, which are designed to provide human-level dexterity, multi-planar flexibility and 360 degrees of articulation.
The arms are controlled by a surgeon from the Hominis Surgical System console, which allows them to replicate the motions and capabilities of a surgeon’s arms, shoulders, elbows and wrist joints.
Memic said the system will be sold at a significantly lower price than its competitors, potentially allowing more facilities to adopt the tool.
Memic CEO Dvir Cohen said: “We are pleased to receive FDA De Novo authorisation of our Hominis system, which offers a small, cost-effective and less invasive option over current robotic instruments limited to straight shaft and single wrist designs and controlled with large, complex and expensive equipment.
“This authorisation is also just the beginning; it opens the door for our novel system to expand to additional indications that, until now, have been off-limits to robot-assisted surgery.”
The company plans to pursue general surgery and transluminal indications for the Hominis platform and is developing artificial intelligence (AI)-enabled features to support its surgical indications.
The FDA will require Memic to develop a comprehensive training program for surgeons and operating room staff to complete before operating the device.
In an FDA-evaluated 30-patient study of the safety and effectiveness of Hominis hysterectomies, all procedures were completed successfully with no conversions to an open-surgery or another laparoscopic approach.
Robot-assisted surgery has become increasingly popular during the Covid-19 pandemic, as it allows procedures to be performed remotely, without touching the patient.
Robotic surgery can also offer significant clinical benefits to patients, such as less pain and scarring, shorter recovery times and lower infection rates.
In October 2020, Da Vinci surgical robot manufacturer Intuitive Surgical announced the launch of a new venture capital fund to invest in early-stage companies supporting minimally-invasive procedures. Disrupted surgical schedules as a result of Covid-19 impacted the company’s financial performance in 2020.



