Scientists based at EPFL’s Institute of Bioengineering in Lausanne, Switzerland have simulated aspects of embryo formation inside a microfluidic chip, setting the stage for fabricating functional tissues and organs for drug testing and transplantation.
In a developing embryo, stem cells receive a highly dynamic range of concentrated signalling molecules called morphogens, which tell the pluripotent stem cells what kind of specialised tissues and cells to become.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
One issue clinicians face when attempting to construct tissues in vitro is how to present morphogens to the cultured cells, where the time and dose is vital.
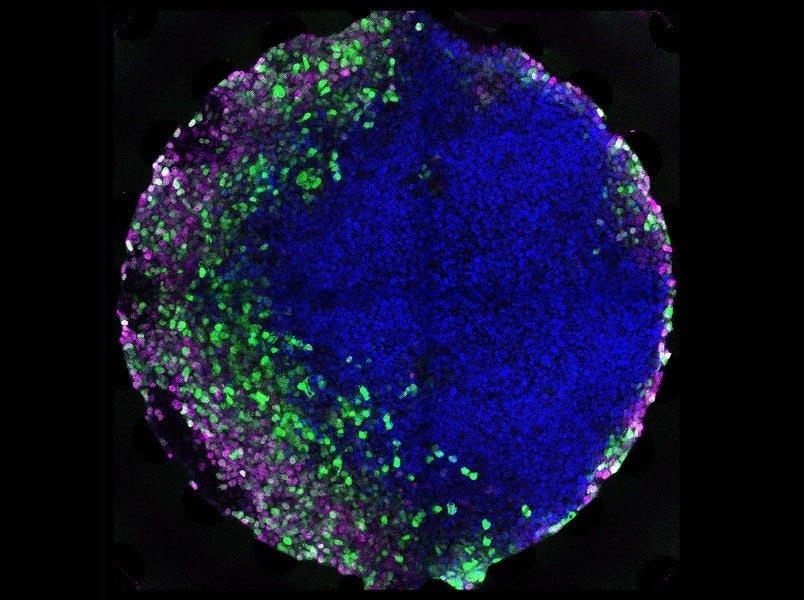
The EPFL team discovered that this could be mitigated by growing the stem cells in a microfluidic device, a chip with small channels that allow for the precise control of tiny amounts of fluid. Using these channels, they were able to expose the stem cells to very carefully controlled concentration gradients of various morphogens.
EPFL professor Matthias Lütolf said: “Simply exposing a collection of stem cells to a single concentration of a morphogen ends in uncontrolled morphogenesis because the cells lack important instructions.”
The results of the controlled microfluidic morphogen exposure were impressive: the cells developed and organised themselves into domains of different cells types depending on the concentration of morphogens they were exposed to, just like they do in the body.
They were able to successfully mimic aspects of gastrulation, an early stage of embryonic development, paving the way for growing specific human tissues in the lab in a more controlled manner.
EPFL scientist Dr Andrea Manfrin said: “We hypothesized that engineering an artificial signaling center ‘ex vivo’ could allow us to steer the self-organization of a stem cell population towards a desired outcome. This has obvious advantages for tissue and organ engineering.”
Stem cells grown on the microfluidic chip in this way could provide the opportunity to develop new tools for drug testing and regenerative medicine. It could also enable scientists to study processes related to developmental biology and provide alternatives to animal experimentation in certain areas of research.
Lütolf, who is already working with groups at the Lausanne University Hospital and beyond to generate miniaturised organs from patient-derived cells, said: “One of our long-term goals is to engineer organs for transplantation. We are still far from growing functional organs in a dish; but recent progress in stem cell biology and bioengineering make me optimistic that this can become a reality. The key is to better understand how cells themselves build tissues and organs in the embryo”.





