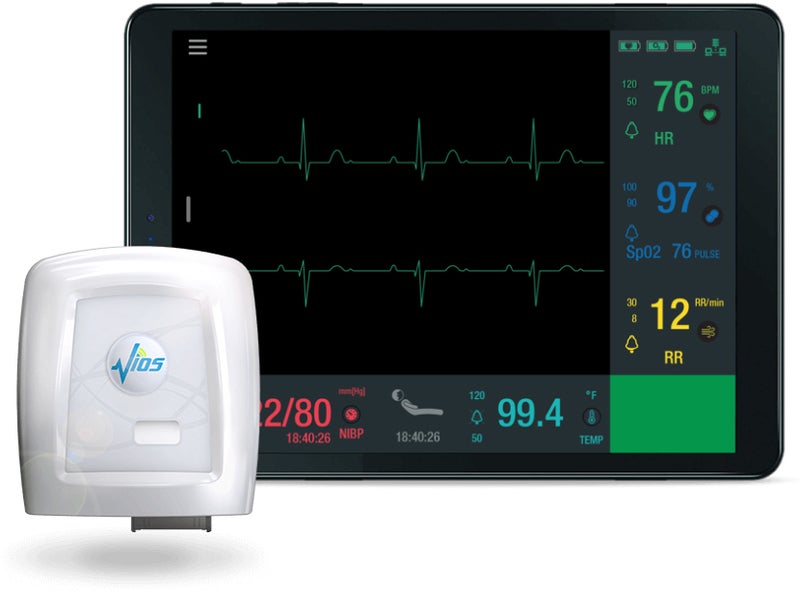
Murata Vios has received clearance from the US Food and Drug Administration (FDA) for the Vios Monitoring System (VMS).
The approval of the company’s two 510(k) submissions for the second-generation wireless suite of products will provide a platform designed to reduce the cost of patient-monitoring equipment.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The user-friendly system monitors patients throughout various clinical environments by gathering 7-leads of ECG, heart rate, respiratory rate, SpO2, blood pressure, temperature, pulse rate, patient posture and activity data.
Murata Vios senior manager Varun Verma said: “Our FDA clearance is a big step in our commitment to patient safety and providing a premium patient experience.
“The US healthcare landscape is rapidly evolving, and we look forward to providing efficient and secure solutions that help manage patients throughout the care continuum.”
The system is capable of continuously monitoring and displays a patient’s vital signs across a range of devices that use sensors, unique signal processing IP, and software that runs on standard off-the-shelf hardware.
Healthcare providers can use the low-bandwidth connected data to remotely monitor patient data in near real-time.
A subsidiary of Murata Manufacturing, Murata Vios plans to launch the platform in the US and is currently working with hospitals and post-acute care facilities in preparation.
Murata Vios CEO Amit Patel said: “Our team has worked long and hard to achieve this milestone and we are excited to finally bring our solution to market.
“Now that we have been acquired by Murata Manufacturing of Japan, we will be able to support and scale our solution in ways that very few innovative technologies can.”





