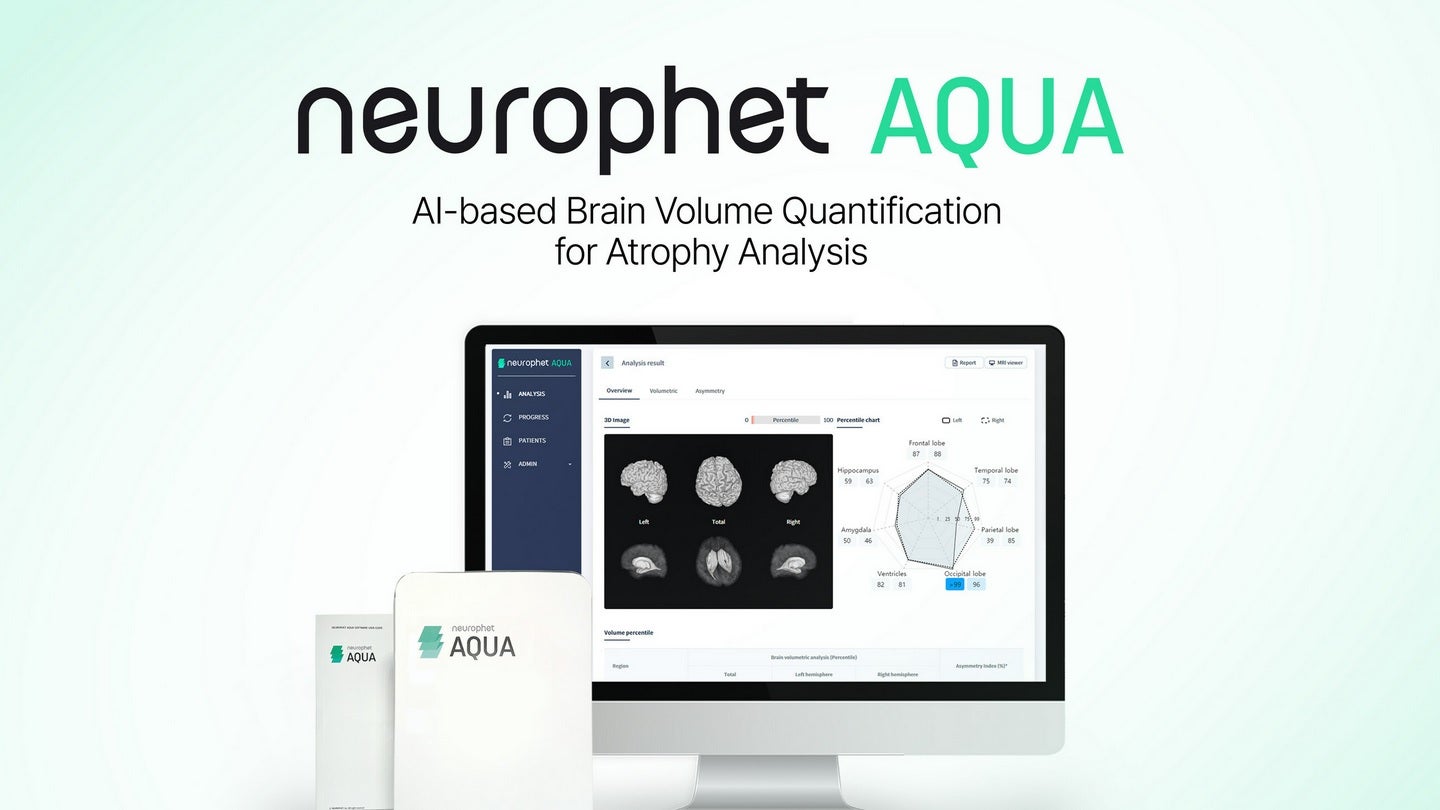
Neurophet has secured 510(k) clearance from the US Food and Drug Administration (FDA) for its brain magnetic resonance imaging (MRI) analysis software, Neurophet AQUA.
The new software is intended for the analysis of brain atrophy noticed in neurodegenerative diseases such as Alzheimer’s disease.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Neurophet AQUA, the company’s flagship product, has the ability to segment and analyse brain images within five minutes irrespective of gender, race or age.
In August last year, the company also received 510(k) clearance from the FDA for its brain positron emission tomography (PET) scan analysis software, Neurophet SCALE PET.
Neurophet CEO Jake Junkil Been said: “Since Neurophet AQUA is FDA cleared, CE marked and has received medical device approval from Japan, Singapore and Korea, we plan to supply our product to the global market through active sales activities.
“FDA has cleared the technology of Neurophet AQUA to analyse brain MRI and Neurophet SCALE PET to analyse brain PET scan which determines the prescription for Alzheimer’s disease treatments, it is expected to be used essentially for clinical trials and the prescription of Alzheimer’s disease treatments.”
Neurophet intends to collaborate with companies offering radiology artificial intelligence (AI) solution platform services to distribute its products in the US market.
At present, the company is engaged in negotiations with related companies regarding the distribution of Neurophet AQUA. The company develops AI-powered solutions for diagnosis support, treatment devices and treatment guides for brain diseases.
The company also offers brain imaging treatment planning software, Neurophet tES/TMS LAB, for electric and magnetic brain stimulation, as well as the cloud-based brain imaging analysis research tool Neurophet SegPlus.





