Researchers at Imperial College London have developed a new technology that can aid in grading tumour biopsies, eliminating human subjectivity and variability in determining cancer severity.
Named Digistain, the new technology is expected to aid in better diagnosis and treatment of cancer.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
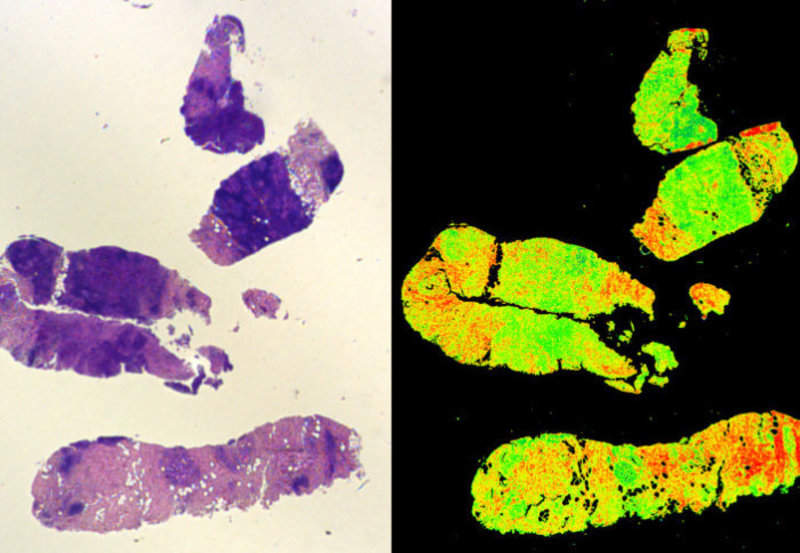
At present, cancers are commonly diagnosed using a biopsy thinly sliced and stained with two vegetable dyes. The ‘H+E’ stained sample is observed under a microscope and the tumour severity is graded by eye.
However, various doctors analysing the same biopsy slice will only agree on the severity grade approximately 70% of the time, leading to over-treatment of some patients.
The new Digistain technology is designed to employ invisible mid-infrared light to measure a biological marker called the nuclear-to-cytoplasmic-ratio (NCR) factor, which is known to be present in several cancers.
Imperial College London Physics department professor Chris Phillips said: “Our machine gives a quantitative ‘Digistain index’ (DI) score, corresponding to the NCR, and this study shows that it is an extremely reliable indicator of the degree of progression of the disease.
“Because it is based on a physical measurement, rather than a human judgement, it promises to remove the element of chance in cancer diagnosis.”
The team tested the technology in a double-blind clinical trial using two adjacent slices obtained from 75 breast cancer biopsies.
One of the slices was graded with the H+E staining and used to locate the ‘region of interest’ (RoI), area of the slice having the tumour.
Subsequently, Digistain was used to obtain a DI score averaged over the corresponding RoI on the other slice and statistical analysis was carried out on the results, which revealed significant correlation of DI score with the H+E grade.
Phillips added: “The strength of this correlation makes us extremely optimistic that Digistain will be able to eliminate subjectivity and variability in biopsy grading.”
The team believes that Digistain could be easily and cheaply integrated into hospital laboratories and would be easy to validate by comparing with numerous existing biopsy specimens.





