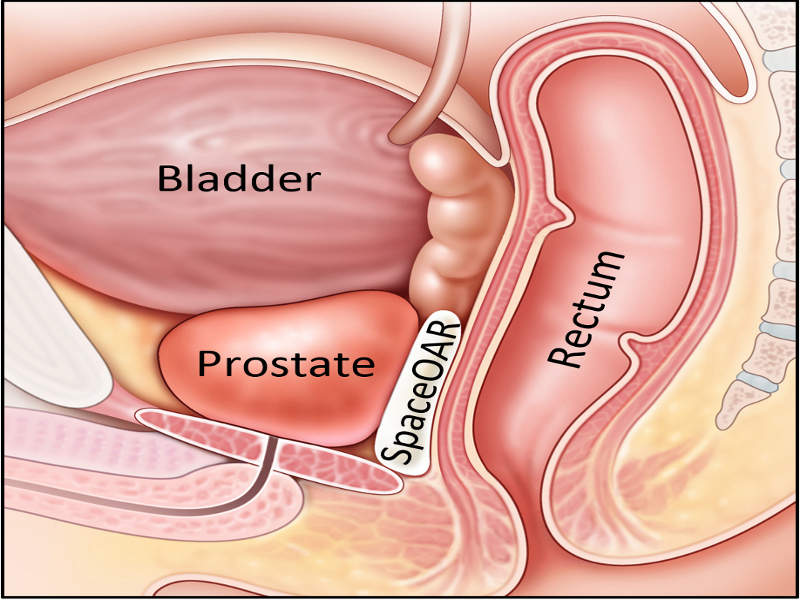

US-based medical technology company Augmenix has reported positive long-term data of its Phase III clinical trial of the SpaceOAR System to separate the rectum and prostate during prostate cancer radiotherapy.
SpaceOAR System has been developed to temporarily separate the anterior rectal wall from the prostate during radiotherapy, limiting the exposure of the anterior rectum to radiation.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Radiation therapy for prostate cancer poses a risk for the adjacent healthy tissue which might cause a range of bowel, urinary symptoms that can affect patient health and quality of life during radiotherapy, and in the future.
The system is injected as a liquid into the space between the prostate and rectum where it solidifies into a soft hydrogel and remains for three months, it eventually liquefies and is completely absorbed after radiation treatment.
The Phase III trial was conducted as a prospective, randomised, multi-centre, patient-blinded clinical study, which tested the effect of radiation on critical organs like the prostate, rectum, bladder, penile bulb, rectile and urinary toxicity and quality of life (QOL) in prostate cancer patients with and without SpaceOAR System.
Results suggested a 73.5% reduction in rectal V70 radiation dose, and a 49% reduction in median penile bulb radiation dose in patients treated with SpaceOAR (SpaceOAR) compared to patients who did not receive SpaceOAR hydrogel (Control).
After three years of treatment, the SpaceOAR group experienced 78% reduction in late rectal toxicity complications, compared to control patients.
Results also indicated a 75% reduced risk of mild urinary incontinence among patients treated with SpaceOAR compared to the control group.
The average SpaceOAR patient bowel and urinary QOL measure was intact as it was before radiotherapy, while the Control patients QOL measures had declined.
The SpaceOAR patients experienced 2.5% QOL decline compared to a 20% decline of the control patients.
Augmenix CEO John Pedersen said: “Following radiation therapy, patients often experience a decline in urinary, bowel and sexual function which in some cases can be extreme.
“The data shows that the benefits seen in the use of SpaceOAR are durable and support the use of SpaceOAR hydrogel spacing during radiotherapy for prostate cancer with the potential to help many patients return to and maintain pre-treatment quality of life.”
Image: A diagram showing the placement of SpaceOAR. Photo: courtesy of Business Wire.





