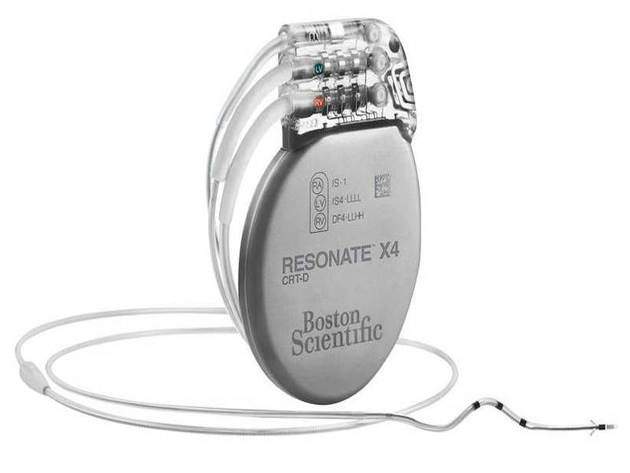

Boston Scientific has released its new Resonate implantable cardioverter defibrillator (ICD) and cardiac resynchronisation therapy defibrillator (CRT-D) devices with the HeartLogic Heart Failure Diagnostic for heart failure (HF) management.
The US Food and Drug Administration (FDA) has approved the systems for conditional use in the magnetic resonance imaging (MRI) environment.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Designed to alert physicians of worsening HF, the new HeartLogic Diagnostic combines data from sensors used for heart sounds, respiration rate and volume, thoracic impedance, as well as heart rate and activity.
The alert feature is said to have demonstrated 70% sensitivity with the ability to deliver weeks of advance notice of an upcoming HF event.
Boston Scientific Rhythm Management and Global Health Policy senior vice-president and chief medical officer Kenneth Stein said: "The HeartLogic Diagnostic provides physicians with the ability to pivot from reactive heart failure treatment to proactive care with a goal of improving patient outcomes and reducing heart failure-related hospitalisations.”
Developed to improve patient management with personalised care, the defibrillators feature the firm’s SmartCRT with EnduraLife Battery Technology.
Using the Multisite Pacing capability for multi-electrode pacing, SmartCRT Technology allows customised use and placement of the device.
In combination with EnduraLife Battery, SmartCRT enables personalisation of the device based on individual patient needs.
The MR-conditional labelling of the devices facilitates full-body MR scans of patients implanted with these devices in 1.5 Tesla environments upon compliance with conditions of use.
Boston Scientific is currently enrolling patients in the MANAGE-HF study designed to further assess the HeartLogic Diagnostic.
The firm is also conducting various clinical trials to investigate improved CRT therapy response with the SmartCRT Technology.
Image: All Resonate devices feature the HeartLogic Heart Failure Diagnostic that could provide weeks of advance notice of an impending HF event. Photo: courtesy of Boston Scientific Corporation.



