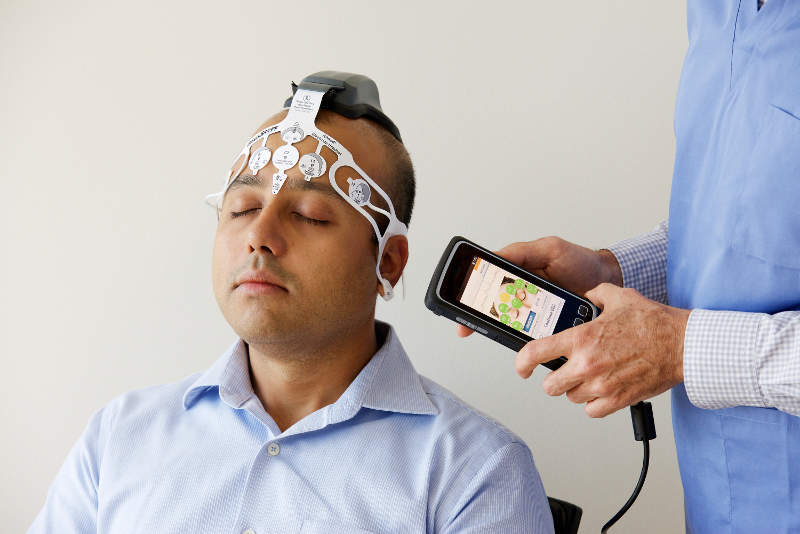

US-based medical neurotechnology company BrainScope has released its Ahead 300 product and is planning its limited launch among a selected group of leaders through its Centers of Excellence programme.
Developed in partnership with the US Department of Defense, the Ahead 300 facilitates rapid and objective assessment of mildly presenting head-injury across the full spectrum of brain injury at the point of care for up to three days after incidence of injury.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It determines the likelihood of a mild head injury leading to a traumatic structural brain injury and detects anything that is functionally abnormal with the brain after a head injury.
The Ahead 300 device uses handheld technology and a proprietary disposable sensor headset.
It also features BrainScope’s patented electroencephalography (EEG) capabilities which use signal processing, sophisticated algorithms and machine learning to aid in examination of head-injured patients with objective biomarkers of brain electrical activity.
BrainScope CEO Michael Singer said: “Following an eight year evolution of development, which included 4 FDA clearances, we are announcing today the launch of the Ahead 300 for sale.
“The Ahead 300 offers a multi-modal, comprehensive capability to address a vast market need for the epidemic of mild head injury in this country.
“We know the absence of such a capability in current medical practice is a great concern for clinicians, as well as for head injured patients and their loved ones.
“There is now available, for the first time, an FDA cleared medical device the Ahead 300 which can help the clinician objectively and rapidly assess and disposition patients, right at the point of care.”
The Ahead 300 product comprises four objective tests and 16 standard concussion assessment tools which can be customised into a configurable panel on the Ahead 300 handheld device.
Image: The Ahead 300 facilitates rapid, objective assessment of mild head injury. Photo: courtesy of Businesswire.



