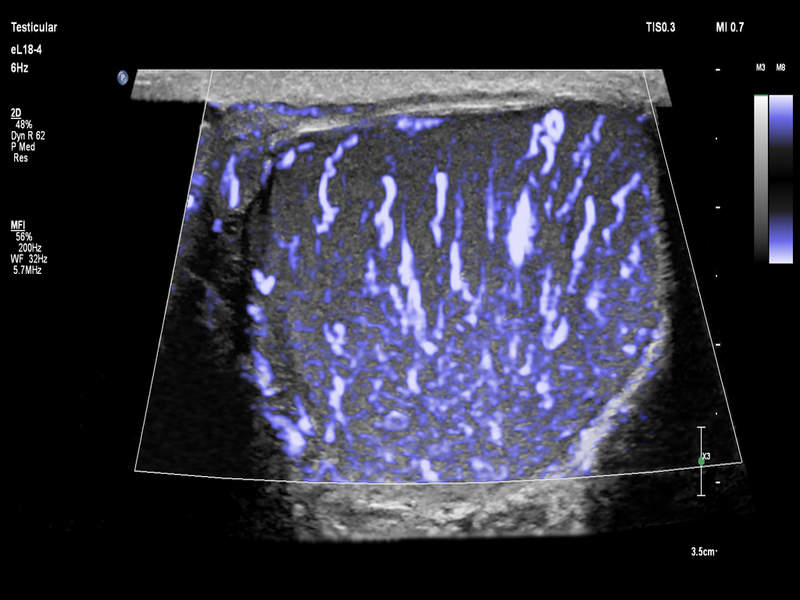
The US Food and Drug Administration (FDA) has granted 510(k) clearance to Royal Philips’ new eL18-4 PureWave linear array transducer, which offers a complete solution for the detection of abnormalities in small organs close to the skin through an ultrasound exam.
The Philips Ultimate Small Parts Solution includes MicroFlow Imaging, Elastography, and Precision Biopsy.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Available on Philips EPIQ 7 and 5 and Affiniti 70 ultrasound systems, the small parts assessment solution can be used for imaging diseases and disorders of small organs, including breasts, testicles and thyroid. It can also be used to detect musculoskeletal injuries such as sprains and tears.
Philips Ultrasound business leader Vitor Rocha said: “The superb image quality derived from Philips’ breakthrough, ultra-broadband frequency transducer, along with full solution Elastography support, helps clinicians make confident clinical decisions driven by an exam that is easier to perform.
“With the new Philips Ultimate Small Parts Solution, clinicians now have the power to comprehensively assess and treat small parts and deliver better care for their patients with all-in-one functionality.”
The eL18-4 transducer is said to deliver high-quality detail resolution and tissue uniformity for various clinical solutions such as bowel, paediatrics, vascular and obstetrics, while the Philips MicroFlow Imaging is designed to assess small vessel blood flow.
Philips Elastography solution features strain and ElastQ Imaging shear wave capability to provide definitive information on tissue stiffness, while Precision Biopsy is developed to reduce needle blind zones and aid in improving the display of needle reflections.
The new solution is intended to allow confident diagnosis, maximum productivity, connected workflow and patient data security.





