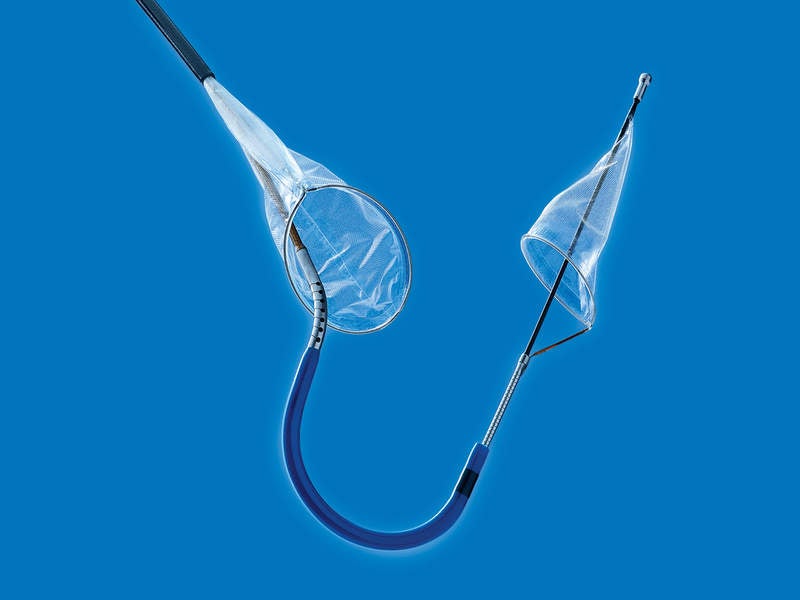

The US Food and Drug Administration (FDA) has granted de novo clearance to Claret Medical for commercialising its Sentinel cerebral protection system (CPS).
Developed to provide protection from the risk of stroke, the new CPS is designed to capture and remove debris ejected during transcatheter aortic valve replacement (TAVR) before it reaches the brain.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The data from a randomised controlled SENTINEL clinical trial indicated a 63% decrease in strokes during the first 72 hours following TAVR, while there was also a substantial difference at 90 days.
The new device is set to launch at select high-volume TAVR centres of excellence across the country.
SENTINEL trial clinical steering committee chairman Martin Leon said: “The fact that one in four patients had an average of 25 particles of debris collected that were visible to the naked eye is striking.
“The device was delivered safely, added minimal time to the procedure, and performed as intended with capture of embolic material and reduction in ischemic brain injury.”

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataIn 99% of TAVR cases, the device was found to eliminate visible debris that headed towards the brain, with no added risk, regardless of the replacement valve type.
The CPS enables positioning away from the aortic arch, which results in decreased interference with TAVR catheters, no damage to the aortic arch, while it allows physicians to focus on the procedure.
Claret Medical president and CEO Azin Parhizgar said: “By helping to reduce the occurrence of periprocedural strokes, we intend to provide patients and physicians with greater peace of mind as they approach a TAVR procedure.”
The Sentinel was assessed in more than 1,000 patients and has been used to protect more than 3,500 patients across the world.
Image: The Sentinel cerebral protection system. Photo: courtesy of Claret Medical Inc.





