
Medical device company Lumendi has obtained the US Food and Drug Administration (FDA) 510(k) clearance for its endoscopic accessory, DiLumen, for complete positioning of an endoscope in the large intestine and assisting with optical visualisation, diagnosis and endoscopic treatment.
DiLumen has been designed to stabilise a colonoscope to better facilitate incision-free endolumenal therapeutic procedures, which are complex procedures involving usage of flexible colonoscope and therapeutic devices inserted into the colon to track and treat affected tissue while preserving the colon.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The device has been developed by Lumendi in collaboration with the Minimally Invasive New Technologies programme (MINT) at Weill Cornell Medicine and New York-Presbyterian.
It is based on Lumendi’s Endolumenal Interventional Platform MINT’s initiative which enables the introduction of endolumenal procedures in conducting many gastrointestinal surgeries.
MINT Programme co-director Dr Peter Johann said: “DiLumen’s use during flexible colonoscopy is an important technical advance in a field that has previously been defined by laparoscopic and open surgical procedures.
“DiLumen can stabilise a section of colon and facilitate the endoscopic removal of complex adenomas or polyps, with the potential to positively impact patient outcomes.”
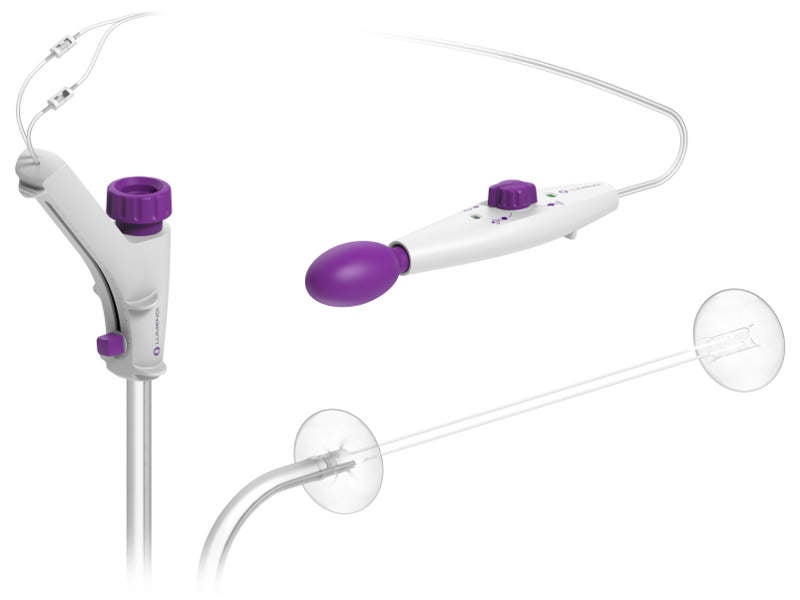
DiLumen features a single-use, soft flexible sheath that fits over standard and small-diameter colonoscopes.
It uses two balloons, one of which is used behind the bending section of the colonoscope and the second in front of the tip of the colonoscope.
The balloons deflates and creates a stable therapeutic zone (TZ) which facilitates more localised insufflation and manipulation of the colon providing improved access to lesions to enable endoscopists and surgeons to perform precise endolumenal interventions.
After completion of the procedure, the balloons are deflated and removed along with the colonoscope.
Image: DiLumen. Photo: courtesy of Business Wire.





