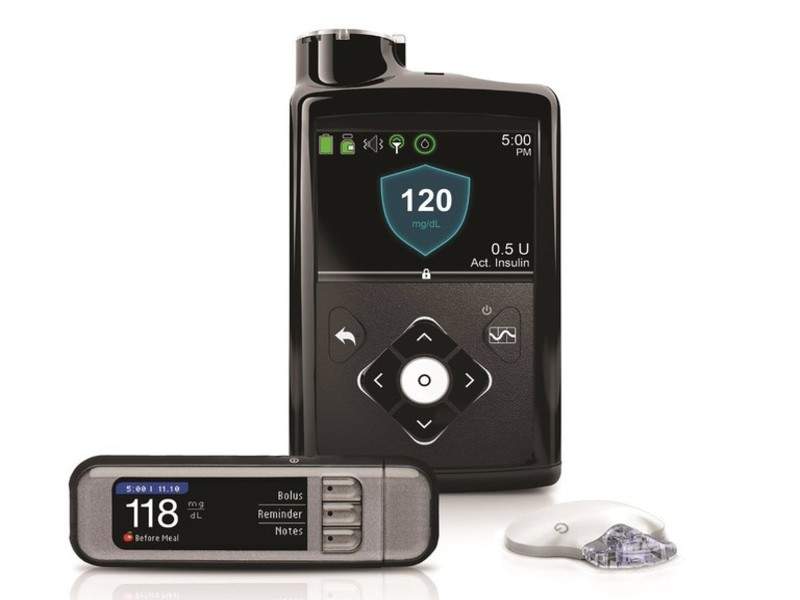

Medtronic has entered a new outcomes-based agreement with US healthcare firm Aetna to investigate health outcomes in diabetes patients switching to pump therapy using its insulin pump that employs SmartGuard Technology and the new MiniMed 670G system.
The solution is designed to self-adjust to maintain patients blood sugar levels according to their personal requirements.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The study will include type 1 and type 2 diabetes patients who are currently on several daily insulin injections for diabetes management.
In addition to the MiniMed 670G system, the agreement will provide Aetna members with access to Medtronic's other advanced diabetes technologies and support services.
Medtronic Diabetes Group president Hooman Hakami said: "Our goal is to continue to lead by driving innovation that demonstrably improves patient outcomes, elevates patient experience, and lowers the total cost of care.
“The growing body of clinical evidence demonstrating the benefits of our proprietary SmartGuard Technology is compelling and we are pleased to work with Aetna to drive awareness and align incentives around the technologies that make the biggest difference for patients and for the health system."
The deal also includes Medtronic's reimbursement depending on clinical improvement thresholds for Aetna members switching to the insulin pump.
Medtronic Diabetes Group Americas group vice-president said: "This agreement is an important first step as we look to broadening our partnership to facilitate patient access to the most advanced diabetes management solutions across the care continuum that not only ensure outcomes, but lower the overall cost of care for this chronic and burdensome disease."
Image: MiniMed 670G system. Photo: courtesy of Medtronic plc.





