Medtronic has reported that the first patients have been randomised in Symplicity HTN-4, the first randomised clinical trial to investigate renal denervation for the treatment of moderate uncontrolled hypertension in US patients.
The study will assess the Symplicity renal denervation system in patients with moderate uncontrolled hypertension, with systolic blood pressure greater than or equal to 140mm Hg and less than 160mm Hg, despite treatment with three or more anti-hypertensive medications of different classes.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Symplicity HTN-4 randomised its first patients at Medical University of South Carolina (MUSC), Piedmont Heart Institute and Duke University Medical Center. It is Medtronic’s second randomised, controlled renal denervation clinical trial in the US.
Symplicity HTN-4 builds upon the Symplicity HTN-3 study, Medtronic’s pivotal US clinical trial of the Symplicity renal denervation system.
Medtronic Renal Denervation vice president and general manager Nina Goodheart said: “Symplicity HTN-4 demonstrates Medtronic’s commitment to providing randomised safety and efficacy data for renal denervation in a wide variety of patients, as well as helping increase our understanding of the potential benefit of renal denervation for more patients with treatment resistant hypertension.”
Symplicity HTN-4 will enrol up to 580 patients at approximately 100 sites, continuing to target a patient population in line with the uncontrolled hypertension defined by the Joint National Committee on the Prevention, Detection, Evaluation and Treatment of High Blood Pressure (JNC-7), the American Heart Association and the European Society of Hypertension.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataSimilar to the US pivotal trial, Symplicity HTN-3 study evaluated patients with uncontrolled hypertension with a systolic blood pressure greater than or equal to 160mm Hg. The Symplicity HTN-4 study will be blinded and include a sham control.
The principal investigators of Symplicity HTN-4 are Dr David Kandzari, director and chief scientific officer of Interventional Cardiology and Interventional Cardiology Research, Piedmont Heart Institute, Atlanta, and Dr Michael Weber, professor of medicine, Division of Cardiovascular Medicine, at the SUNY Downstate College of Medicine in Brooklyn, New York.
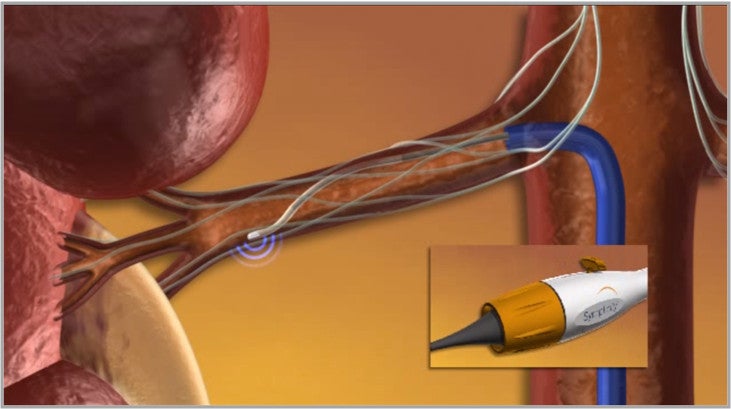
The Symplicity renal denervation system consists of a proprietary generator and flexible catheter, which delivers a controlled, low-power radio-frequency (RF) energy through a proprietary algorithm, or pattern, aiming to deactivate the surrounding renal nerves.
While the Symplicity system had received CE mark approval in 2008 it is currently available for only investigational use in the US.
Image: The Symplicity renal denervation system. Photo: courtesy of Medtronic Inc.





