US-based Medtronic has revealed the details of six-month analysis of first 1,000 patients enrolled in the Global Symplicity Registry, the largest renal denervation registry in the world.
The results of this observational, open-label study demonstrated that renal denervation with the Symplicity renal denervation system resulted in significant reductions in both office and 24-hour ambulatory blood pressure compared to baseline, with very low adverse event rates to six months.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Of the 1,000 patients enrolled at the time of the analysis, six-month safety data was available for 913 patients.
The overall office systolic blood pressure reduction was 11.9mm Hg (n=751) and 24-hour ambulatory systolic blood pressure reduction was 7.9mm Hg (n=404).
For patients with office blood pressure >= 160mm Hg and ambulatory blood pressure >= 135 at baseline receiving greater >= 3 antihypertensive medication classes had a blood pressure reduction of 20.2mm Hg at six months (n=244).
University of Saarland, Homburg/Saar, Germany chairman, Department of Internal Medicine and Global SYMPLICITY Registry co-chair Dr Michael Böhm said the Global SYMPLICITY Registry provides further evidence confirming the well-established safety profile of renal denervation with the Symplicity system.
"Presented on the heels of Symplicity HTN-3, this real-world experience provides a significant contribution to the discussion about the efficacy of renal denervation and offers additional perspective when considering the option of renal denervation in clinical practice for high-risk patients suffering from uncontrolled hypertension who may have limited treatment options," Dr Böhm said.
On March 29, the company announced the full results of the Symplicity HTN-3 clinical trial, which were presented in late-breaking session at ACC and published simultaneously in The New England Journal of Medicine.
Symplicity HTN-3 is the first and only blinded, randomised, sham controlled study of renal denervation for treatment-resistant hypertension, met its primary safety endpoint but did not meet its primary or powered secondary efficacy endpoints.
The Global Symplicity Registry will enrol a minimum of 5,000 patients who undergo the renal denervation procedure, from more than 200 sites worldwide with planned follow-up to five years.
The registry also will also document the data for other diseases characterised by elevated sympathetic drive, such as diabetes mellitus type 2, heart failure and chronic kidney disease.
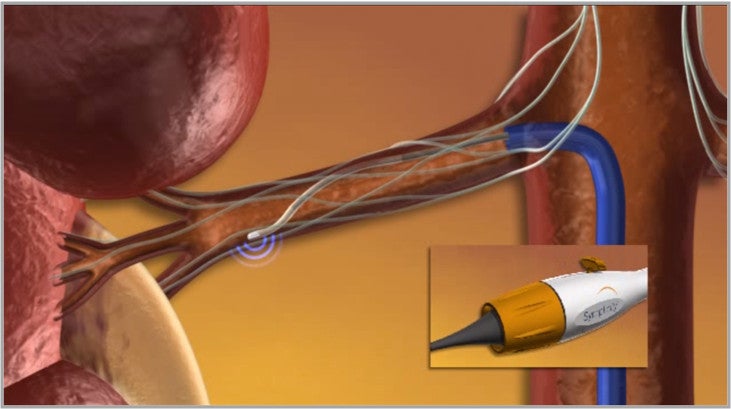
The Symplicity renal denervation system consists of a proprietary generator and flexible catheter, which delivers a controlled, low-power radio-frequency (RF) energy through a proprietary algorithm, or pattern, aiming to deactivate the surrounding renal nerves.
The Symplicity system received CE mark approval in 2008 and is currently available for investigational use only in the US.
Medtronic anticipates that its global Symplicity clinical programme will involve more than 8,000 patients including the US, Europe, and Japan, with more than 1,200 of these patients participating in randomised clinical trials.
Image: Symplicity Renal Denervation System. Photo: courtesy of Medtronic Inc.





