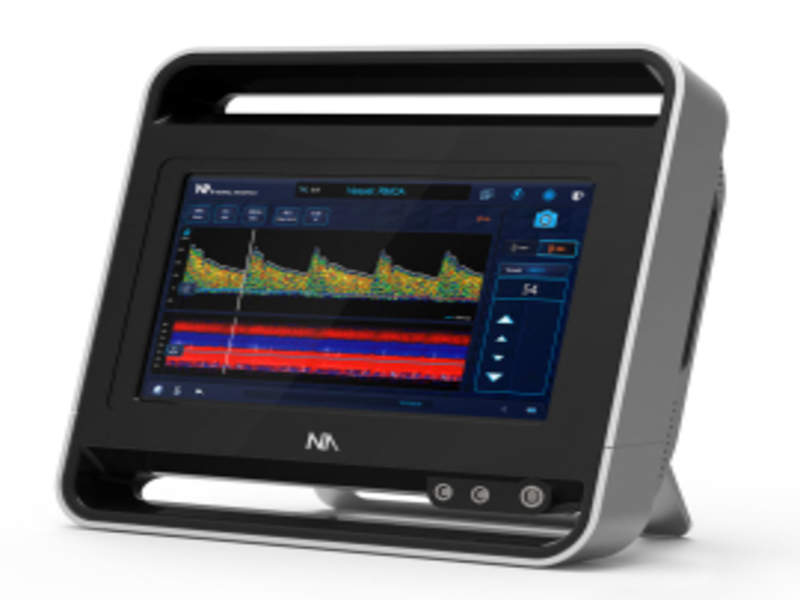

Medical device company Neural Analytics has secured CE mark for its ultrasound device Lucid M1 Transcranial Doppler Ultrasound System (Lucid System) to diagnose and monitor patients with brain disorders.
A battery operated medical grade tablet device, the Lucid System is equipped with Transcranial Doppler, an ultrasound which externally evaluates the blood vessels in the brain.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The new system avoids the necessity for invasive tests and can be used from a physician’s office to diagnose brain disorders, primarily caused due to disruption of the blood flow.
The firm intends to immediately commence the commercial selling of the Lucid System in Europe.
Neural Analytics chief executive officer Leo Petrossian said: “The CE Mark for our Lucid System represents an important milestone for Neural Analytics as it will allow us to bring a non-invasive device to patients with blood flow disorders in Europe who may benefit from this portable monitoring system.”
With the US Food and Drug Administration (FDA) 510(k) clearance obtained in last October, the system has received an International Standards Organisation (ISO) 13485:2003 and ISO 13485:2003 CMDCAS certification along with CE mark.
Founded in 2013, Neural Analytics combines data science with hardware to develop products and services which measure, diagnose and monitor brain health.
The firm’s portable and autonomous devices are claimed to be reliable as well as generate precise and objective physiological measurements for medical responder monitoring of neural disorders.
Image: The Lucid M1 Transcranial Doppler Ultrasound System. Photo: courtesy of Neural Analytics.





