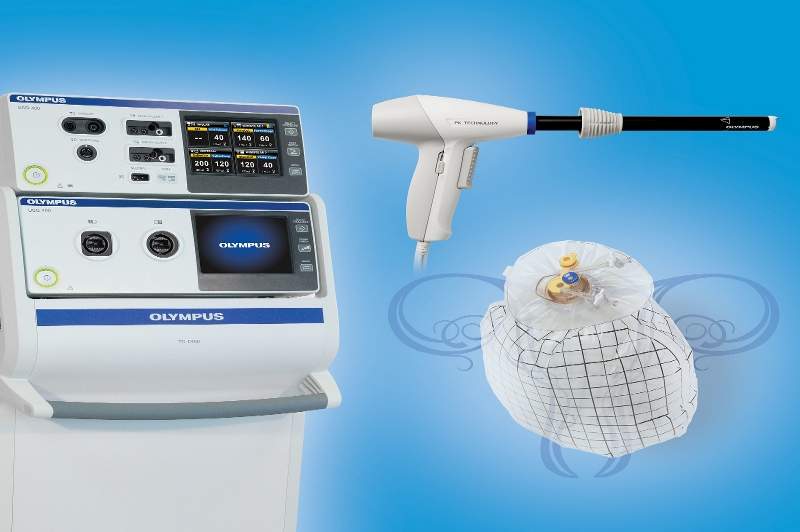

Olympus America has unveiled its Contained Tissue Extraction System for laparoscopic tissue containment and extraction.
The Contained Tissue Extraction System features the PneumoLiner, a US Food and Drug Administration (FDA)-cleared containment device for gynecologic surgery and the PK Morcellator which has recently obtained the FDA 510(k) clearance.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The laparoscopic PK Morcellator has been designed to provide certain patients with a laparoscopic surgery option to avoid open hysterectomy and myomectomy for uterine fibroid removal.
Sloane Women’s Hospital vice-chair and chief of gynecology Arnold Advincula said: “Contained tissue extraction is continually evolving.
“As surgeons, it is our duty to evaluate potential solutions to the surgical challenges we face in clinical practice.
“We are pleased that Olympus has taken the initiative to develop a unique and well-tested system designed specifically to facilitate the option for a laparoscopic approach in women who are at a low-risk of having an unexpected malignancy at the time of surgery.”
The Olympus Contained Tissue Extraction System allows surgeons to isolate and perform uterine tissue morcellation and extraction using the PneumoLiner containment device which has obtained approval to be used with certain laparoscopic power morcellators to isolate uterine tissue that is not suspected to contain cancer.
After being insufflated, the PneumoLiner allows space and vision throughout the power morcellation procedure while preventing the escape of fluids, cells, and tissue fragments.
However, both the PneumoLiner containment device and PK Morcellator have not been approved to be used in women with tissue that is known or suspected to contain malignancy and cannot be used for removal of uterine tissue containing suspected fibroids in patients who are peri or post-menopausal, or candidates for en bloc tissue removal vaginally or via mini-laparotomy.
Image: The Olympus Contained Tissue Extraction System. Photo: courtesy of Olympus America.





