
Following clearance from the US Food and Drug Administration (FDA), SI-Bone has launched the new iFuse-3D Implant to promote bone growth.
Developed using the firm’s 3D-printing technology, the new titanium implant is designed for use in the sacroiliac (SI) joint.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The triangular titanium design of the iFuse-3D is based on SI-Bone’s iFuse Implant that has been used in more than 26,000 procedures since 2009.
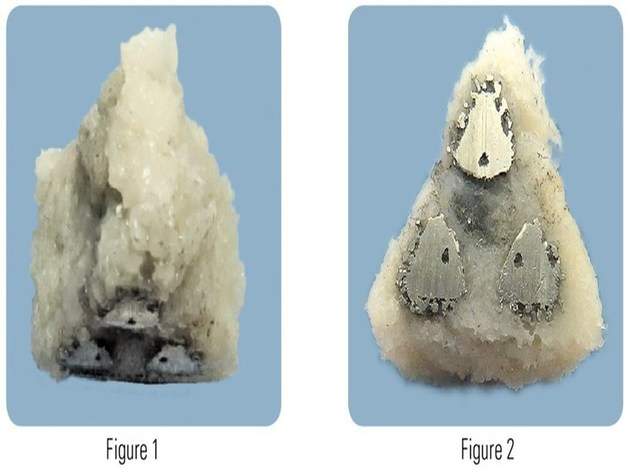
The new implant’s characteristic fenestrations and increased porous surface that is similar to the trabecular structure of cancellous bone are expected to enable bone ongrowth, ingrowth and through growth.
Additionally, iFuse-3D’s surface and structure facilitate increased osteointegration and promote intra-articular fusion.
SI-Bone chief technology officer Scott Yerby said: "The design and development of the iFuse-3D implant was a multi-year effort.
“Our goal was to expand the iFuse family using 3D-printing technology to provide enhanced surface characteristics while retaining key performance features of the iFuse Implant, including superior rotational resistance, mechanical strength, and ease of use with our existing instrumentation.
“iFuse-3D, with its trabecular-like surface, provides 250% greater surface area than our highly successful iFuse Implant. Additionally, the structural fenestrations allow complete bone through growth."
The iFuse Implant System is developed to enable minimally invasive surgical fusion of the SI joint, using triangular titanium implants to establish an interference fit within the ilium and sacrum.
The triangular implant shape and press fit insertion technique allows instant fixation by reducing the joint's motion called nutation.
Image: Implant's porous surface (Figure 1) and its fenestrations (Figure 2). Photo: courtesy of PRNewsfoto/SI-BONE Inc.





