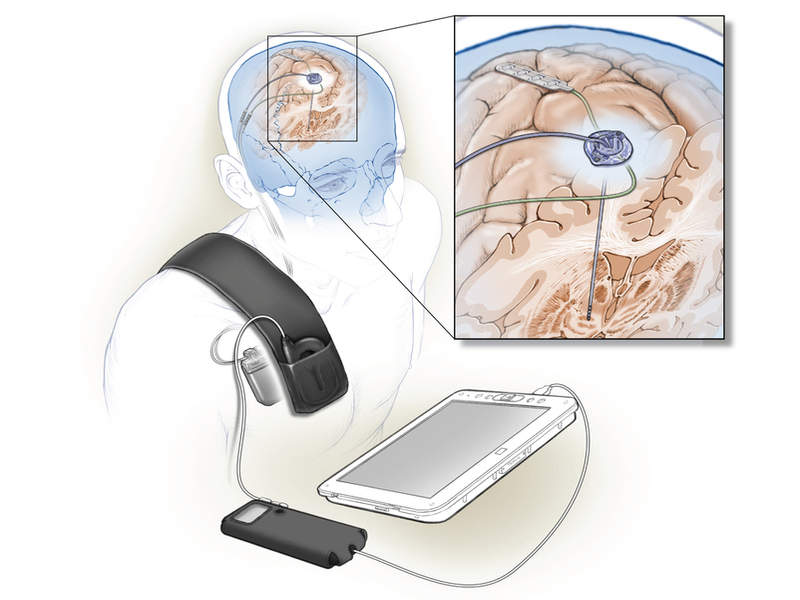
A new study funded by the National Institutes of Health (NIH) BRAIN Initiative has evaluated the first fully implanted deep brain stimulation (DBS) system that self-tunes signalling based on the brain’s feedback and can be used to treat Parkinson’s disease symptoms.
DBS involves the surgical implantation of an electrode into the brain to deliver constant stimulation to the basal ganglia. This standard approach is associated with certain side effects and needs reprogramming by a trained clinician.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The new technique fine-tunes itself in order to provide stimulation which is responsive to a patient’s brain signals in real time. It can monitor as well as modulate brain activity.
In this technique, an electrode is placed over the primary motor cortex and the signals captured are transmitted to a computer programme incorporated in the device to facilitate a decision on stimulation.
BRAIN Initiative team lead Nick Langhals said: “The novel approach taken in this small-scale feasibility study may be an important first step in developing a more refined or personalised way for doctors to reduce the problems patients with Parkinson’s disease face every day.”
For their short-term study, the researchers trained the computer programme to identify a pattern of brain activity associated with dyskinesia, a DBS side effect in Parkinson’s, as a guide to deliver stimulation.
Two subjects were implanted with the adaptive DBS device, which decreased the stimulation upon detection of dyskinesia-related brain activity and vice-versa in order to reduce the side effects.
The study was designed to investigate the feasibility and effectiveness of this adaptive DBS approach and found the device to be equally effective at controlling symptoms when compared with standard DBS.
While there was no improvement in movement, the self-tuning device minimised usage of the device’s battery.
The researchers intend to further validate the device’s effectiveness in larger, longer-term trials to manage symptoms of Parkinson’s disease patients.





