US-based medical device company Nuvaira has raised fresh funds in an equity financing round led by US Venture Partners to further develop its lung denervation system for treating chronic obstructive pulmonary disease (COPD) and asthma.
The $79m financing round was also joined by Endeavour Vision, Qiming Venture Partners, Lightstone Ventures and the Richard King Mellon Foundation.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Nuvaira’s existing investors including Advanced Technology Ventures, Morgenthaler Ventures, Split Rock Partners, Versant Ventures, Vertex Venture Holdings and Windham Venture Partners also participated in the latest funding round.
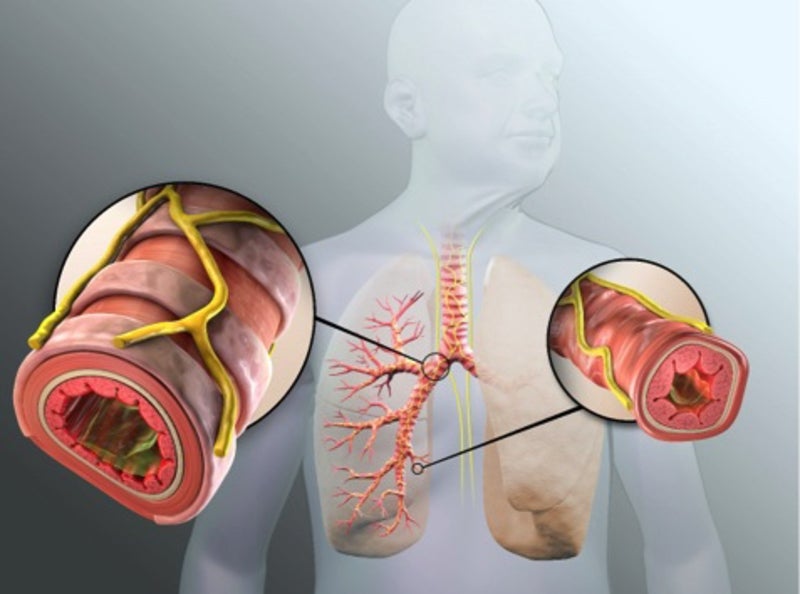
Nuvaira Lung Denervation System is a catheter-based system designed to address the airway hyper-responsiveness in COPD and asthma through a procedure called Targeted Lung Denervation (TLD).
TLD is a one-time bronchoscopic procedure that disrupts overactive nerves in the lungs in order to open up the airways for improved breathing.
The device obtained the European CE-Mark in January 2016. Its safety and feasibility were validated in IPS-I/II, AIRFLOW-1 and AIRFLOW-2 clinical trials.
Nuvaira CEO Dennis Wahr said: “We are thrilled to have such a strong consortium of top-tier investors in this financing, as a clear vote of confidence in our work and the performance of our novel catheter-based system in our Phase IIb randomised, sham-controlled trial in patients with chronic obstructive pulmonary disease (COPD) patients.”
The company intends to use the latest funds to conduct a pivotal AIRFLOW-3 clinical trial. Data from this trial are expected to support approval from the US Food and Drug Administration (FDA).
In addition, the financing will be used for a targeted clinical development strategy in select European markets.
AIRFLOW-3 trial principal investigator Frank Sciurba said: “We are encouraged by the results of the AIRFLOW-2 trial, and we see TLD potentially filling an unmet need for our COPD patients who continue to have exacerbations while on maximal guideline-based pharmacologic therapy.”
Findings from the AIRFLOW-2 trial showed a more than 50% decrease in respiratory adverse events such as COPD exacerbations and hospitalisations.
The World Health Organization has claimed that COPD could be the third leading cause of death in the world by 2030.
Last week, digital therapeutics company Kaia Health has announced that it will undertake a feasibility study to assess the effectiveness of its digital therapeutic COPD treatment in Japan’s ageing population.
Additional reporting by Charlotte Edwards.





