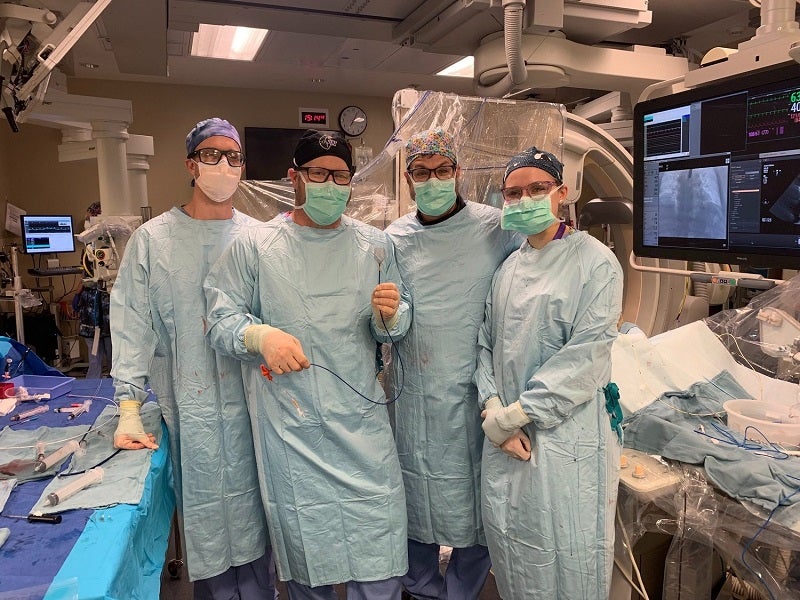
ŌNŌCOR has announced the first-in-human use of the catheter-based ŌNŌ endovascular retrieval system for the percutaneous removal of an intracardiac tumour, a process which normally requires open-heart surgery.
The new retrieval system has been designed for receiving, aligning, compressing and percutaneously removing non-biologic and biologic material from the vascular system.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
ŌNŌCOR stated that the ŌNŌ system is simple to use and compatible with endovascular snares, vascular sheaths and other graspers that are available commercially.
It has been developed to make catheter-based retrieval quick and safe, as well as to eliminate the need for remedial surgical procedures.
At the University of Washington (UW) Medical Center in Seattle, Washington, US, Dr James McCabe and Zachary Steinberg, along with their team, used the ŌNŌ and electrocautery to snare, resect and remove a cardiac fibromyxoma from the right atrium of a 54-year-old patient.
The procedure provides access to a new therapy for heart mass patients across the world.
UW Interventional Cardiology section chief Dr McCabe said: “Intracardiac tumours are a vexing problem and present a management dilemma for physicians. These tumours are often found incidentally by echocardiography at relatively small sizes.
“However, rather than subjecting patients to elective surgical removal, these masses are typically followed expectantly until they increase in size to a point where they are physiologically problematic. At that point, the risks of open-heart surgery are deemed justifiable.”
In May, the ŌNŌ retrieval system received 510(k) clearance from the US Food and Drug Administration (FDA).
The company noted that the device is currently available at a few sites across the US.





