Orlucent has received breakthrough device designation (BDD) from the US Food and Drug Administration (FDA) for its skin fluorescence imaging system.
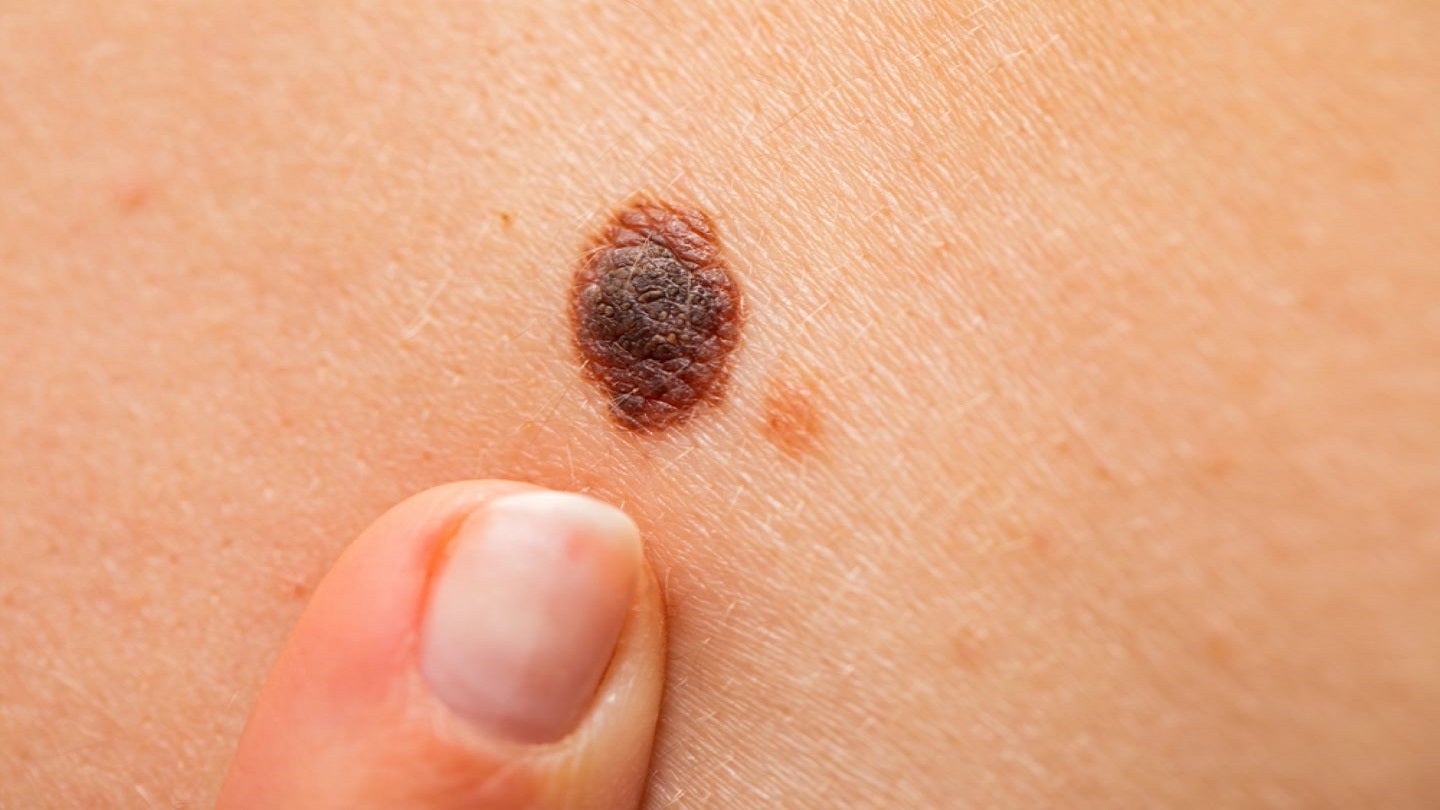
The Orlucent system is a handheld point-of-care molecular-level mole imaging and analysis device.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It can detect and evaluate biological tissue-remodelling activity related to the formation of unusual moles in adults, without the need for invasive procedures.
Physicians can use the system alongside conventional clinical visual skin examination, as well as information on the medical history and skin type of the patient.
The system will aid physicians in establishing a clinical course of action and detecting melanoma in its earliest stages.
Orlucent president Dr Catherine Shachaf said: “It’s estimated that anywhere from 2% to 53% of US patients have moles that may be classified as suspicious, atypical or dysplastic and the Orlucent system will be an important tool to provide their physicians with information on a mole’s biological activity status as well as it’s potential to become melanoma.”
There are three steps to evaluate an atypical lesion using the Orlucent system. Firstly, a healthcare professional administers the reagent and wipes it away. The healthcare professional subsequently deploys the imager to capture images under both white light and fluorescence
In the third step, the image analysis software of the system displays and analyses the images. The software provides a probability score that indicates the likelihood of a suspicious mole undergoing tissue remodelling.
The Orlucent system’s score, combined with a physician’s clinical assessment, helps determine whether to remove a mole for biopsy or continue observing it for potential future retesting.





