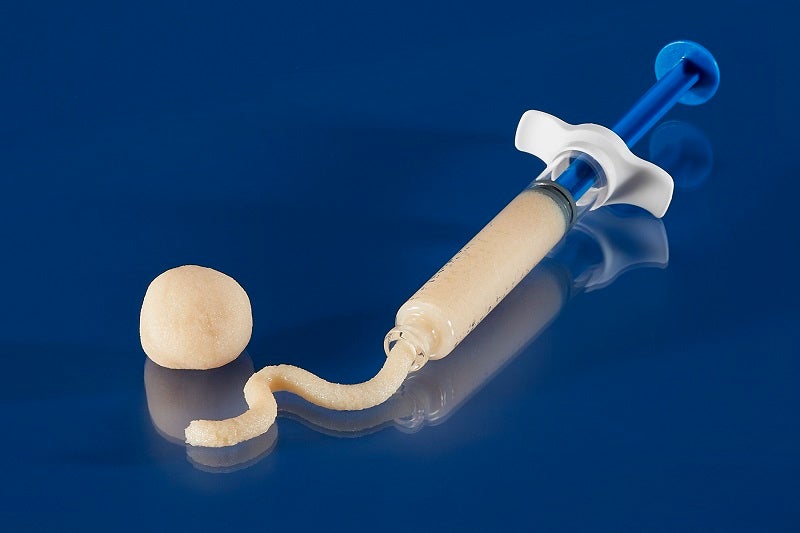
Orthofix Medical has announced the complete commercial release and first patient implant of its Legacy Demineralized Bone Matrix (DBM).
The Legacy DBM is a putty which has been developed to fill voids or gaps in boney defects or traumatic injuries of the spine, pelvis, or extremities.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It has a putty-like consistency and can be used as an extender with allograft or autograft, or with bone marrow aspirate.
Non-profit organisation MTF Biologics has processed the Legacy DBM using its specialised processing techniques that maintain the inherent growth factors present in bone.
University Orthopedics Spine Surgery chief Dr Alan Daniels has implanted the Legacy DBM in the first patient.
Dr Daniels said: “Bone healing is a dynamic process where multiple factors play an integral role in deciding which biologic best meets the individual needs of the patient to aid them on their road to recovery.
“Legacy DBM provides a cost-effective option, without compromising clinical experience or the handling needed in a bone matrix.”
Orthofix Medical noted that Legacy DBM joins its recently introduced FiberFuse Advanced and FiberFuse Strip allografts, Trinity Elite, Virtuos Lyograft, and the AlloQuent structural allograft portfolio.
The company also sells and distributes the OpusMg Set bone void filler, the O-Genesis graft delivery system, Opus One osteoconductive scaffold, and Opus BA bioactive bone graft.
Orthofix Global Spine president Kevin Kenny said: “The Legacy DBM is another example of our continued commitment to expanding biologic offerings to meet the needs of surgeons and their patients.
“In the past year alone, we have introduced five new biologics, including the Virtuos Lyograft, a first-of-its-kind, shelf-stable, and complete autograft substitute.
“We understand there are unique surgeon users for a wide range of bone-grafting options, and it is our goal to be a leading provider of these biologic offerings to our customers.”





